Caelifera
Shorthorned Grasshoppers, Locusts and Relatives
Hugh Rowell and Paul Flook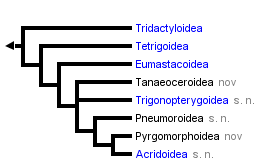


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Recent estimates (Kevan 1982; Günther, 1980, 1992; Otte 1994-1995; subsequent literature) indicate some 2400 valid Caeliferan genera and about 11000 valid species described to date. Many undescribed species exist, especially in tropical wet forests. The Caelifera are predominantly tropical, but most of the superfamilies are represented world wide.
By insect standards the Caelifera are relatively homogenous; all have jumping back legs and are almost exclusively herbivorous. None the less they show considerable diversity. As adults they range in size from a few millimetres to more than 15 cm in length, are flighted or flightless, occupy virtually all non-marine habitats in which plants can live (including deserts, water surfaces, the crowns of forest trees, grasslands, or underground); they eat algae, mosses, the leaves and reproductive organs of ferns, gymnosperms and angiosperms, or even the roots of the latter, with all degrees of foodplant specialisation from wide-range polyphagy to strict monophagy.
The Caelifera are probably the oldest living group of chewing folivorous insects. The Tettigonioidea may predate them geologically, but it is unclear how herbivorous these were, as many Tettigonioids are even now still carnivorous or omnivorous. The fossil Caelifera are reviewed by Zeuner (1941-1944), Sharov (1971), Kukalova-Peck (1991), Carpenter (1992) and Ross & Jarzembowski (1993). The split between the Caelifera and the Ensifera is not more recent than the Permo-Triassic boundary (Zeuner 1939). The earliest known representatives of an extant Caeliferan Superfamily are the extinct *Regiatidae (Tridactyloidea) from the Lower Jurassic (Gorochov 1995). Essentially modern Eumastacids are known from the mid-Jurassic, modern Tridactyloids and Tetrigoids from the early Cretaceous, Acridoids from the Eocene. Most, possibly all, of the modern superfamilies probably developed in the Jurassic.
Characteristics
Among other synapomorphies the Caelifera is distinguished from the Ensifera by the structure of the ovipositor, in which the original 6 valves are reduced to 4 functional ones with transverse musculature, by antennae composed of less than 30 segments, and by the absence of auditory organs on the prothorax - if a tympanum or other hearing organ is present, it is abdominal. The sperm are thin and elongate, with an acrosome inserted on the nucleus by means of two lateral processes.
Discussion of Phylogenetic Relationships
A conservative view of caeliferan relationships is shown in Figure 4. Due to the virtual absence of explicit schemes in the modern literature, this hypothesis follows no particular publication, but it is shared by most modern morphologists (Rentz, 1991).
================== Tridactyloidea
===|
| =============== Tetrigoidea
===|
| ============ Eumastacoidea
===|
| ========= Trigonopterygoidea
===|
| ====== Pneumoroidea
===|
| === Pamphagoidea
===|
=== Acridoidea
Figure 4: Phylogeny based on morphology, modified after Dirsh (1975).
As in several of the insect orders, few formal phylogenetic analyses of the Caelifera have been undertaken. The major sources of phylogenetic opinion lie in the Discussion sections of primarily taxonomic or morphological publications, and are rarely supported by explicit data. Cladistic analysis of morphological data matrices is rare. Only recently have molecular data been used to examine phylogenetic relationships within this group.
One category of phylogenies is derived from wing venation, as exemplified by the schemes of Zeuner, Sharov, Ragge and Gorochov. In these works, however, the emphasis is on the Ensifera, which have a much more extensive fossil record, and the Caelifera are not treated in detail. Further, some of the Caeliferan relationships implicit in the scheme proposed by Sharov are not supported by any obvious evidence (see e.g. the critique in Amédégnato, 1993). A further problem lies in the identification of the plesiomorphic form of the venation. For example, Ragge concluded that the Pneumorids were the most primitive Caeliferans on the basis of the similarity of their venation with that of the Palaeozoic *Palaeodictyoptera. However, as these are not considered to be ancestral to the orthopteroid insects, their significance in this respect is dubious.
A second category of phylogeny is derived primarily from the male internal genitalia (e.g. Roberts, Dirsh, Descamps, Amédégnato). The most rigorous analysis of genital characters for large numbers of taxa (Amédégnato 1977) was based on phenetic similarity rather than cladistic methodology, and the original data matrix is not available. The resultant phylogenies are however generally in agreement with those derived from molecular data. Numerical phenetic analysis has also been applied to non-genital morphological characteristics to produce a phylogeny of the Orthopteroid orders (Blackith & Blackith 1967), but the number of Caelifera included in the sample was very small.
Molecular approaches to higher level Caeliferan systematics consist to date of a) cladistic analysis of allozyme polymorphism of glycolytic enzymes (Colgan, 1989), again using a very small taxonomic sample, and b) parsimony and distance analysis of a much larger sample of mitochondrial and nuclear ribosomal sequences by the present authors (Flook and Rowell 1997-2000). Perhaps surprisingly, and reassuringly, most of these diverse methodologies produce phylogenies in rough agreement with each other. Originally we analysed the different ribosomal gene sequences separately. Two molecular phylogenies so derived are indicated below (the phylogenies are consensus trees based on parsimony, distance matrix and maximum likelihood reconstructions).
====== Tridactyloidea =============== Tridactyloidea
| |
===|===== Tetrigoidea | ============ Tetrigoidea
| | | |
| ====== Eumastacoidea | | ========= Eumastacoidea
<<===| | | |
| ====== Pneumoroidea sensu Dirsh <<===| | | ====== Pneumoroidea sensu Dirsh
| | ===| | |
===| === Pamphagoidea sensu Dirsh ===| |===== Trigonopterygoidea sensu Dirsh
===| ===|
=== Acridoidea |===== Pamphagoidea sensu Dirsh
|
====== Acridoidea
12S + 16S mt rRNA gene tree 18S rRNA gene treeFigure 5: Molecular phylogeny of Caelifera after Flook and Rowell (1997a, 1998)
The topologies differ in the levels at which they resolve, but the branching order of the Superfamilies is not contradicted in either phylogeny. The mitochondrial data do not resolve well the oldest branches, presumably due to site saturation, whereas the 18S data do not resolve well the youngest branches, due to insufficient divergence. For this reason we subsequently used combinations of these data sets, with appropriate precautions, to achieve a broader resolution. This strategy has now allowed us to resolve the phylogeny of the entire Caelifera at the level of Superfamily.
The resultant tree is the following:
|==================== Tridactyloidea
|
<<===| |================= Tetrigoidea
| |
| | |============== Eumastacoidea
|==| |
| | |=========== Tanaeoceroidea nov
|==| |
| |=========== Trigonopterygoidea s.n.
|==|
| |======== Pneumoroidea s.n.
|==|
| |==== Pyrgomorphoidea nov
|===|
|==== Acridoidea s.n.
Figure 6: Combined 18S, 12S and 6S gene tree (Flook et al., 1999, 2000)
These molecular data often support unequivocally one of several schemes previously proposed on morphological grounds. Thus, for example, the basal placing of the Tridactyloids, the relatively close relationship of the Tetrigoids and Eumastacoids, and the placement of the Trigonopterygoids as a separate group between the Eumastacoids and Pneumoroids, have all been previously suggested by some (though not all!) morphologists, and will probably be generally accepted without dissent.
Among our more original findings is the dismemberment of the Pneumoroidea sensu Dirsh, with the resultant superfamily status of the Tanaoceroidea, the placement of the Xyronotidae as the sister family of the Trigonopterygidae and their joint elevation to superfamily status as the Trigonopterygoidea, and the restriction of the Pneumoroidea sensu novo to the sole family Pneumoridae. These results are supported by very high levels of statistical probability. The data do not as yet allow resolution of the branching order of the Tanaeoceroidea and Trigonopterygoidea.
The other important finding concerns the Pamphagoidea sensu Dirsh. Some morphologists have seen this group as part of the Acridoidea, others considered the differences in genitalia to warrant separate superfamily status; it was however generally accepted that a) they are monophyletic or at least closely related and b) they are primitive with respect to the remaining Acridoidea. The ribosomal sequences however indicate that this group is actually polyphyletic; the Pyrgomorphidae is indeed a monophyletic clade branching off well before the Acridoidea, but the remaining taxa (principally Pamphagidae & Lentulidae) are embedded within the Acridoidea. We have formalized this finding by raising the Pyrgomorphidae to superfamily status, dropping the superfamily Pamphagoidea and redefining the Acridoidea s.n. to include the Pamphagidae & Lentulidae (see 'A Classification of the Caelifera'). The allozyme data of Colgan (1989), which included a pyrgomorphid, are compatible with our proposition, but do not confirm it, as no other pamphagoid sensu Dirsh was included. Recently, however, Eades (2000) has offered new interpretations of the phallic anatomy of pyrgomorphids, pamphagids and acridids which support the new classification.
Classification
The age of the Caelifera and their multiple convergence to common habitats make their morphological classification difficult below the level of the superfamily. For the past 50 years most weight has been placed on the internal genitalia, especially those of the male sex. These are effectively never available in fossil material: the palaeontological classifications, as in most insect orders, are founded principally on wing venation (Kukalova-Peck, 1991).
Modern authors have usually divided the living Caelifera on morphological grounds into 7 higher taxa, which we here treat as superfamilies (see Figure 4 above). The majority of living Caelifera belong to the family Acrididae of the Acridoidea. The difficulties in Orthopteran taxonomy have been discussed by Rentz (1991, p. 378) who remarks: "There are many disparate classifications of the orthopteroid insects (or positions thereof) at the present time. The overriding theme is the escalation of the rank of categories above the tribal level. The lack of congruence among authors contributes to considerable instability. Readers should consult Dirsh (1975) for the extreme of these views and Kevan (1982) for a synthesis of the classifications." The most disputed superfamilies have traditionally been the Pamphagoidea, which some authorities regard as insufficiently separated from the Acridoidea to merit independent status, and the Eumastacoidea, which may or may not include the family Proscopiidae. The position of these taxa is discussed in the Discussion of Phylogenetic Relationships, and the accessory page 'A Classification of the Caelifera' provides a proposal for a classification down to the subfamily level which takes in account the results of recent morphological and molecular analyses.
Additionally, there are two Permian/Mesozoic fossil caeliferan taxa, the *Locustopseidae and the *Locustavidae, each sometimes given Superfamily rank, but by others (e.g. Gorochov 1995) grouped as Superfamily *Locustopsoidea. These families are defined only on the basis of wing venation (trifurcate MA and a branched MP+CuA1 in the forewing, branched MA in the hindwing). At least the former family may be simply an assemblage of primitive forms and contain the early representatives of several of the modern groups.
Other Names for Caelifera
- Shorthorned grasshoppers
- locusts
- caeliferans
- Shorthorned Grasshoppers, Locusts and Relatives
References
Amédégnato, C. (1974). Les genres d'Acridiens neotropicaux, leur classification par familles, sous-familes et tribus. Acrida 3: 193-204.
Amédégnato, C. (1976) Structure et évolution des genitalia chez les Acrididae et familles apparentées. Acrida 5: 1-15
Amédégnato, C. (1977) Etude des Acridoidea Centre et Sud Americains (Catantopinae sensu lato): Anatomie des genitalia, classification, repartition, phylogenie. Thesis, Université Pierre et Marie Curie, Paris. 385 pp. (mimeo.)
Amédégnato, C. (1993) African-American relationships in the Acridians (Insecta, Orthoptera). In: "The Africa-South America connexion" (W. George & R. Lavocat, Eds.), pp. 59-75, Oxford University Press, Oxford.
Ander, K. J. (1939) Vergleichend anatomische und phylogenetische Studien über die Ensifera (Saltatoria). Opuscula Ent. suppl. II, Lund, 8, 306 pp.
Blackith, R. E. and R. M. Blackith (1968) A numerical taxonomy of orthopteroid insects. Austr. J. Zool. 16: 111-131.
Carpenter, F. M. (1992) "Treatise on invertebrate palaeontology. Part R: Arthropoda 4. Vol 3 & 5: Superclass Hexapoda", University of Kansas Press, Lawrence, Kansas.
Chopard, L. (1920) "Recherches sur la conformation et la développement des derniers segmentes abdominaux des Orthoptères". Thèse Faculté des Sciences de Paris, Oberthur, Rennes. 352 pp., pl. 7.
Colgan, D. (1989) Developmental variation and allozymic polymorphism in the enzymes of glycolysis in grasshoppers. J. evol. Biol. 2: 141-151.
Descamps, M. (1973) Revision des Eumastacoidea aux échelons des familles et des sous-familles (genitalia, repartition, phylogénie). Acrida 2: 161-298
Dirsh, V. M. (1956) The phallic complex in Acridoidea (Orthoptera) in relation to taxonomy. Trans. R. ent. Soc. Lond. 108: 223-356
Dirsh, V. M. (1975) "Classification of the acridomorphoid insects", Farringdon, Oxford, Classey.
Eades, D.C. (2000) Evolutionary relationships of phallic structures of Acridomorpha (Orthoptera). Journal of Orthoptera Research 9: 181-210.
Flook, P. K. & Rowell, C. H. F. (1997a) The phylogeny of the Caelifera (Insecta, Orthoptera) as deduced from mitochondrial rRNA gene sequences. Molecular Phylogenetics & Evolution 8: 89-103.
Flook, P. K. & Rowell, C. H. F (1997b) The effectiveness of mitochondrial rRNA gene sequences for the reconcstruction of the phylogeny of an insect order, Orthoptera. Molecular Phylogenetics & Evolution 8: 177-192.
Flook, P. K. & Rowell, C. H. F. (1998) Inferences about orthopteroid phylogeny and molecular evolution from small subunit nuclear ribosomal RNA sequences. Insect Mol. Biol. 7: 163-178.
Flook, P. K. Klee, S., & Rowell, C. H. F. (1999) A combined molecular phylogenetic analysis of the Orthoptera and its implications for their higher systematics. Syst. Biol. 48: 233-253.
Flook, P. K. Klee, S., & Rowell, C. H. F. (2000) Molecular phylogenetic analysis of the basal Acridomorpha (Orthoptera, Caelifera): resolving morphological character conflicts with molecular data. Mol. Phyl. Evol. 15: 345-354.
Gorochov, A. B. (1995) [Contribution to the systematics and evolution of the order Orthoptera] (Russian) Zool. Journal 74: 39-45
Günther, K. K. (1980). Katalog der Caelifera-Unterordnung Tridactylodea (Insecta). Dtsch. ent. Z. N.F. 27: 149-178.
Kevan, D. K. McE., Akbar S. S. and Chang Y-C. (1969) The concealed copulatory structures of the Pyrgomorphidae (Orth. Acridoidea). Part I. General introduction. EOS Madrid 44: 165-266
Kevan, D. K. McE. (1982) Orthoptera. In "Synopsis and classification of living organisms, Vol 2", (S. P. Parker, Ed.), pp. 352-379, McGraw-Hill, New York.
Kukalova-Peck, J. (1991) Fossil history and the evolution of Hexapod structures. In "The insects of Australia, second edition", (I.D. Naumann, Ed.), Vol. 1, pp. 141-179, Melbourne University Press, Victoria
Otte, D. (1994a) "Orthoptera species file 2. Grasshoppers (Acridomorpha) A. Eumastacoidea, Trigonopterygoidea, and Pneumoroidea". Philadelphia, The Orthopterists' Society and the Academy of Natural Sciences of Philadelphia.
Otte, D. (1994b) "Orthoptera species file 3. Grasshoppers (Acridomorpha) B. Pamphagoidea". Philadelphia, The Orthopterists' Society and the Academy of Natural Sciences of Philadelphia.
Otte, D. (1995a) "Orthoptera species file 4. Grasshoppers (Acridomorpha) C. Acridoidea: Lentulidae, Pauliniidae, Tristiridae, Romaleidae, Acrididae (part)". Philadelphia, The Orthopterists' Society and the Academy of Natural Sciences of Philadelphia.
Otte, D. (1995b) "Orthoptera species file 5. Grasshoppers (Acridomorpha) D. Acridoidea: Acrididae (part)". Philadelphia, The Orthopterists' Society and the Academy of Natural Sciences of Philadelphia.
Rehn, J. A. G., and H. J. Grant. (1958) The phallic complex in the subfamilies of New World Eumastacidae and the family Tanaoceridae. Proceedings of the Academy of Natural Sciences of Philadelphia 110: 301-319, pl. 26-30.
Roberts, H. R. (1941) A comparative study of the subfamilies of the Acrididae (Orthoptera) primarily on the basis of their phallic structures. Proc. Acad. Nat. Sci. Philadelphia 93: 201-246
Ross, A. J. and E. A. Jarzembowski (1993) Arthropoda (Hexapoda; Insecta). In: "The fossil record, 2nd edition", (I. Benton & M. A. Whyte, Eds.), pp. 363-426, London, Chapman and Hall.
Rowell CHF Flook PK (1998) Phylogeny of the Caelifera and the Orthoptera as derived from ribosomal RNA gene sequences. J. Orthopt. Res. 7: 147-156.
Sharov, A. G. (1971) Phylogeny of the Orthopteroidea. (translated from the Russian). Israel Program for scientific translations, Jerusalem. (Translated from Akademiya Nauk SSSR, Trudy palaeontologicheskogo Instituta 118).
Zeuner, H. (1942) The fossil Acrididae (Orth. Salt.) Part II. Oedipodinae. Ann. Mag. nat. Hist. (Ser. 11) 9: 128-134.
Zeuner, H. (1942) The fossil Acrididae (Orth. Salt.) Part III. Acridinae. Ann. Mag. nat. Hist. (Ser. 11) 9: 304-314.
Zeuner, H. (1942) The Locustopsidae and the phylogeny of the Acridodea (Orthoptera). Proc. R. Entomol. Soc. Lond. (Series B) 11: 1-18.
Zeuner, H. (1944) The fossil Acrididae (Orth. Salt.) Part IV. Acrididae incerta sedis and addendum to Catantopinae. Ann. Mag. nat. Hist. (Ser. 11) 9: 359-383.
Title Illustrations

| Scientific Name | Agriacris tricristata |
|---|---|
| Comments | Acridoidea |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1996

|
| Scientific Name | Thericles zebra |
|---|---|
| Comments | Eumastacoidea |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1996

|
| Scientific Name | Tetrigoidea |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1996

|
About This Page

University of Basel, Switzerland

Accelrys, Inc., San Diego, California, USA
Correspondence regarding this page should be directed to Hugh Rowell at
Page copyright © 2001 and
 Page: Tree of Life
Caelifera. Shorthorned Grasshoppers, Locusts and Relatives.
Authored by
Hugh Rowell and Paul Flook.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Caelifera. Shorthorned Grasshoppers, Locusts and Relatives.
Authored by
Hugh Rowell and Paul Flook.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 04 December 1996
- Content changed 23 March 2001
Citing this page:
Rowell, Hugh and Paul Flook. 2001. Caelifera. Shorthorned Grasshoppers, Locusts and Relatives. Version 23 March 2001. http://tolweb.org/Caelifera/13316/2001.03.23 in The Tree of Life Web Project, http://tolweb.org/












 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site