Craniata
Animals with skulls
Philippe Janvier- Hyperotreti (hagfishes)
- Vertebrata (lampreys and jawed vertebrates)
Introduction
The Craniata, or craniates, include all animals having a skull (or cranium, hence their name), be it cartilaginous or bony. A skull is a box of hard tissues which encloses the brain, olfactory organs, eyes, and internal ear. Craniates comprise all fishes - including such jawless fishes as hagfishes and lampreys - amphibians, reptiles, birds and mammals, including Man. The earliest known undisputed craniates are jawless fishes which lived 480 millions years ago. Their evolutionary history took place first in the sea, then in fresh waters. Some craniates, the tetrapods or four-legged vertebrates, became terrestrial and arose about 370 millions years ago from fish ancestors. Now, the majority of the craniate species are represented by one group of fish, the actinopterygians, and the tetrapods. Other craniate groups (jawless fishes, sharks and chimeras, the coelacanth, lungfishes) are considerably depauperate, by comparison with their past diversity.
The Craniata fall into two major clades, the Hyperotreti, or hagfishes, and the Vertebrata. Since the early nineteenth Century, and until recently, the hagfishes were regarded as the sister-group of lampreys (Hyperoartia). The two groups were gathered into the clade Cyclostomi (see below: Discussion of phylogenetic relationships). However, lampreys share with jawed vertebrates, or Gnathostomata, a large number of morphological and physiological characteristics that occur neither in hagfishes, nor in non-craniate chordates (cephalochordates and tunicates). There is, therefore, a rather broad consensus over the theory that hagfishes are the sister-group of the Vertebrata (lampreys and gnathostomes).
Characteristics
The Craniata are characterized by a skull (initially cartilaginous and fibrous), which includes three types of sensory organs derived in ontogeny from ectodermal placodes; that is, thickened patches of the embryonic skin that sink inward toward the brain where they develop into sensory chambers. Anteriormost of these is the olfactory organ, which is initially unpaired, and becomes paired in the Vertebrata. Behind it are the paired eyes, the photoreceptors that develop as lateral outgrowths of the brain. The skin and connective tissues adjacent to the neural (photoreceptive) part of the eye add secondary structures in the Vertebrata (lens, intrinsic muscles, and eye lids). Posteriormost of these sensory organs in the head are the paired acoustic organs or inner ears. The inner ears are mechanoreceptors concerned with hearing, balance, and perception of position of movement. The sensory cells of the inner ear are enclosed in a cavity filled with a liquid, the endolymph, and which develops from one to three semicircular canals. The acoustic organs also comprise a special component, the lateral sensory system, which is lost in most terrestrial craniates (Amniota). It consists of lateralis nerve fibres derived from the acoustic nerve and superficial mechanoreceptors, the neuromasts, which are housed in grooves or canals on the surface of the head. These extend onto the body in the Vertebrata. True neuromasts, however, seem to be unique to the Vertebrata, and have never been observed in hagfishes.

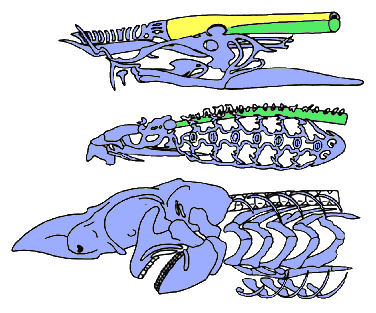
The craniates are characterized by a skull; that is, a complex ensemble of skeletal elements which surrounds the brain and sensory capsules. The skull of hagfishes (top) consists of cartilaginous bars (blue), but the brain is mostly surrounded by a fibrous sheath (yellow) underlain by the notochord (green). The skull of lampreys (middle) has a more elaborate braincase and comprises a large "branchial basket" surrounding the gills. In the gnathostomes (bottom), the braincase is generally closed (after Janvier 1996b).
The skull also encloses the brain, always comprising five parts referred to as the rhombencephalon, metencephalon, mesencephalon, diencephalon, and telencephalon. The metencephalon is developed into a cerebellum in the Gnathostomata and some fossil jawless vertebrates. The nerve fibres are primitively non-myelinated and become myelinated only in the gnathostomes. The brain is continued posteriorly by the spinal cord, which is ribbon-shaped but becomes thicker in the gnathostomes. As in cephalochordates, the dorsal (sensory) and ventral (motor) spinal nerves are initially separate, but unite in the gnathostomes. In all craniates, the olfactory (I), optic (II), trigeminal (V), facial (VII), acoustic (VIII), glossopharyngeal (IX) and vagus (X) cranial nerves are present. Additional cranial nerves, the oculomotor (III), trochlear (IV) and abducent (VI) nerves occur only in the Vertebrata. Some consider that the latter have been secondarily lost in hagfishes.
The olfactory organ opens into a median duct, the nasopharyngeal duct, which also serves the intake of the respiratory water. In most vertebrates, however, this duct becomes a blind tube and the intake of respiratory water is made through the mouth or the gill slits. The nasopharyngeal duct lies ventrally against the diencephalon and there, in ontogeny, induces the formation of an important gland, the hypophysis, or pituitary organ, which comprises neural (neurohypophysis) and glandular (adenohypophysis) parts. The adenohypophysis is particularly complex in the Vertebrata, but very simple in hagfishes.
Craniates possess a unique embryonic tissue, the neural crest, that appears dorsal and lateral to the neural tube and which contributes to a great variety of adult tissues and structures including: sensory neurons (nerve cells), some skeletal and connective tissues in the skull, and some pigment containing cells and other integumentary tissues. In the skull, the neural crest cells give rise to the gill arches, jaws and parts of the braincase floor. In the gnathostomes and a number of fossil jawless vertebrates, the neural crest cells are also involved in the formation of the dermal skeleton (scales, teeth, and dermal bones).
The gills of craniates comprise gill filaments, made up by primary and secondary gill lamellae which insure gas exchanges. In hagfishes, the gills have no skeletal support, and are enclosed in pouches connected to the pharynx. Among vertebrates, a similar structure occurs in adult lampreys only, but here skeletal supports (gill arches) are present. The gills are derived from tissues of the embryonic gut (endoderm), but cells from the embryonic skin (ectoderm) are involved in their formation in the gnathostomes. The respiratory water flow is ensured by a special pumping and anti-reflux organ, the velum, situated at the limit between the mouth and the pharynx. There is a theory that the jaws of the gnathostomes are derived from the velum.
As chordates, all craniates develop a notochord, which is primitively large (hagfishes, lampreys), but becomes transitory in most vertebrates and is replaced by elements of the vertebral column, the centra and arcualia.
All craniates (except most tetrapods) possess a caudal fin strengthened by a number of cartilaginous radials. In vertebrates appear dorsal and anal fins, as well as radial muscles which ensure undulatory movements of the fin web. In the gnathostomes and some fossil jawless vertebrates, there are paired pectoral fins. Only the gnathostomes possess both pectoral and pelvic fins, which are modified into locomotory limbs in tetrapods.
All craniates possess an endoskeleton, which is primitively cartilaginous but becomes mineralized in various ways (bone, calcified cartilage) in the vertebrates. Only the gnathostomes and a number of fossil jawless vertebrates possess a mineralized exoskeleton which develops in the skin tissues. The exoskeleton is made up by a variety of tissues (bone, dentine, enamel).
Craniates have a circulatory system of arteries, capillaries and veins, and a chambered, muscular main heart located ventrally and anteriorly in the trunk. In the Vertebrata, the circulatory system is entirely closed. The two heart chambers, the atrium and ventricle are well apart. There are additional accessory venous hearts in the head and tail, which help in venous blood circulation, but these are lost in the Vertebrata. In gill-breathing craniates, the heart pumps venous blood anteriorly into arteries and capillaries in the gills for gas (oxygen and carbon dioxide) exchange with water. Oxygenated blood then collects dorsal to the gills and flows anteriorly to the head and posteriorly to the organs and muscles, and back to the heart. In some Vertebrata (Osteichthyes) diverticles of the digestive tract (lungs or air bladder) supplements or replaces gills as the repiratory organ.
The digestive tract of craniates is longitudinally differentiated into mouth and oral cavity, pharynx, esophagus, intestine, rectum and anus. A stomach is developed in the Gnathostomata and some fossil jawless Vertebrates. All craniates have a pancreas that produces digestive enzymes and hormones (insulin and glucagon) that regulate blood sugar level. The pancreas was ancestrally disseminated along the anterior part of the gut, but becomes condensed into a well-defined organ in the Vertebrata.
All craniates and the related cephalochordates have a liver or hepatic organ that serves many functions including food storage and production of fat emulsifiers (bile).
The kidneys are the chief excretory organs of vertebrates and these organs play an important role in water and salt balance. Although kidneys vary greatly in size, shape and position among species, all contain nephrons as the basic functional units. Each nephron is a nearly microscopic tubule that receives a filtrate of blood (lacking blood cells and very large molecules). The filtrate is processed by selective secretion and reabsorption of materials to produce an excretory product (generally called urine) that contains nitrogenous waste and other materials. Long and complex kidney tubules occur only in the vertebrates.
Reproductive Biology
The reproductive biology of craniates is highly diverse. The majority of species are bisexual with distinct male and female individuals. Of course, male and female sexes are always distinct in the type of gonads they possess (testes or ovaries), and sex cells they produce (gametes: sperm or ova). Sperm is shed directly into the coelom and then toward the exterior through a pore. In the Gnathostomata, however, testes are linked with the kidneys and sperm pass through the excretory ducts. External sex dimorphism may be non-existent to dramatically pronounced. There are some fishes that are naturally hermaphroditic. In certain hermaphroditic species individuals are "protogynous," i.e. first functioning as females that may later transform into functional males. In other species the opposite sequence of sex change obtains -- "protandrous." There are a few "all-female" species of fishes, amphibians and lizards in which mothers produce only female offspring. In many of these mating with males of related species is necessary to trigger egg development, but fathers do not contribute to the genetic perpetuation of the "all-female" lineages. Among the more typical bisexual craniates there is a very broad spectrum of reproductive mode. The majority of fish and amphibian species are oviparous (egg-laying) with external fertilization of the eggs by the male's sperm. Other fishes, amphibians, many reptiles, all birds and the monotreme mammals (platypus and spiny anteaters of the Australian region) are also oviparous but fertilization is internal. Opposite these are the "live-bearers" or viviparous species in which fertilization is necessarily internal and young develop within the mother's reproductive tract. Here the mother must provide some form nutriment to the embryo (either yolk in the egg or via the blood across permeable placental membranes). Also, the viviparous species have mechanisms for embryonic gas exchange and waste removal. Viviparity has evolved many times in the craniates - among cartilaginous and bony fishes, a handful of amphibians, several snakes and lizards, and most mammals.
Discussion of Phylogenetic Relationships
When initially erected by Linnaeus (1758), the name Craniata was coupled with Vertebrata to include lampreys and all other vertebrates. Hagfishes were, however, regarded as "intestinal worms", yet possibly representing a link between worms and fishes. Dumeril (1806) grouped hagfishes and lampreys (see Hyperoartia) in the taxon Cyclostomi, characterized by horny teeth borne by a peculiar tongue-like apparatus, a large notochord, and pouch-shaped gills. During most of the nineteenth century, the cyclostomes were variously regarded as either degenerate cartilaginous fishes or primitive vertebrates. The latter view, assuming that the cyclostomes are the sister-group of jawed vertebrates has been widely accepted until recently:
Cope (1889) coined the name Agnatha ("jawless") for a group that included the cyclostomes and a number of fossil groups in which jaws could not be observed. From this time on, the vertebrates were divided into two major sister-groups, the Agnatha and the Gnathostomata (jawed vertebrates). The fact that gills were of endodermal origin in the former and of ectodermal origin of the latter was regarded as further support to this classification. As evolutionary ideas progressed, the search for ancestors to the gnathostomes has led some paleontologists to consider that some fossil agnathans (generally the Heterostraci or the Thelodonti) could be more closely related to the gnathostomes than to the cyclostomes or other fossil groups. Consequently, it was more and more widely accepted that agnathans are a grade. As for the living agnathans, the cyclostomes, Stensiö (1927) suggested that they were diphyletic, with hagfishes and lampreys arising separately from two fossil groups of armored agnathans, but retained the opinion that agnathans, as a whole, formed a clade. Løvtrup (1977), however, showed that the characteristics shared uniquely by lampreys and the gnathostomes largely outnumber those shared by hagfishes and lampreys (cyclostome characteristics), thereby suggesting that the cyclostomes are paraphyletic. In other words, the cyclostome characteristics (e.g., horny teeth, "tongue", gill pouches) are primitive craniate characteristics, lost or modified in the gnathostomes. Since only lampreys and the gnathostomes possess vertebral elements, or arcualia, and since hagfishes, lampreys and the gnathostomes all possess a skull, Janvier (1978) proposed to use Linnaeus' initial names Vertebrata and Craniata for the two nested taxa defined by these respective characteristics:
There remains the question of the relationships of the numerous groups of fossil "agnathans" (see, for review, Janvier 1996). Until recently, it was currently admitted that lampreys formed a clade, the Cephalaspidomorphi, with two fossil taxa, the Anaspida and Osteostraci, with which they share the dorsal position of the nasohypophysial opening (external opening of the blind nasopharyngeal duct). Since these fossils possess a well ossified skeleton and paired fins, it was believed that lampreys have lost the exoskeleton and paired fins (Janvier 1978, Forey 1984). Other armored fossil agnathans (e.g. Heterostraci) were thought to be the sister-group of the Cephalaspidomorphi and Gnathostomata, or of the Gnathostomata alone. With the progress of computerized phylogenetic analyses, and the consideration of a large number of characteristics, more parsimonious theories arose, which group with the gnathostomes all fossil agnathans possessing a mineralized exoskeleton (Gagnier 1993, Forey and Janvier 1994). Therefore, all these fossil taxa formerly referred to as "ostracoderms" are treated on the vertebrate page (Vertebrata).
References
Blieck, A. (1992). At the origin of chordates; Géobios, 25, 101-113.
Forey, P. L. (1984). Yet more reflections on agnathan-gnathostome relationships. Journal of Vertebrate Paleontology, 4, 330-343.
Forey, P. L., and Janvier, P. (1993). Agnathans and the origin of jawed vertebrates. Nature, 361, 129-134.
Forey, P. L., and Janvier, P. (1994). Evolution of the early vertebrates. American Scientist, 82, 554-565.
Gagnier, P. Y. (1993a). Sacabambaspis janvieri, Vertébré ordovicien de Bolivie. 1, Analyse morphologique. Annales de Paléontologie, 79, 19-69.
Gans, C. (1989). Stages in the origin of vertebrates: analysis by means of scenarios. Biological Reviews, 64, 221-268.
Gans, C. (1993). Evolutionary origin of the vertebrate skull. In The skull (ed. J. Hanken and B. K. Hall), Vol. 2, pp. 1-35. The University of Chicago Press.
Gans, C. and Northcutt, R.G. (1983). Neural crest and the origin of vertebrates: A new head. Science, 220, 268-274.
Gee, H. (1996). Before the backbone. Chapman & Hall, London.
Hardisty, M. W. (1982). Lampreys and hagfishes: Analysis of cyclostome relationships. In The Biology of Lampreys, (ed. M. W. Hardisty and I. C. Potter), Vol.4B, pp. 165-259. Academic Press, London.
Janvier, P. (1981). The phylogeny of the Craniata, with particular reference to the significance of fossil 'agnathans'. Journal of Vertebrate Paleontology, 1, 121-159.
Janvier, P. (1993). Patterns of diversity in the skull of jawless fishes. In The skull (ed. J. Hanken and B. K. Hall), Vol. 2, pp. 131-188. The University of Chicago Press.
Janvier, P. (1996a). The dawn of the vertebrates: characters versus common ascent in current vertebrate phylogenies. Palaeontology, 39, 259-287.
Janvier, P. (1996b). Early vertebrates. Oxford Monographs in Geology and Geophysics, 33, Oxford University Press, Oxford.
Jefferies, R. P. S.(1986). The ancestry of the vertebrates. British Museum (Natural History), London.
Løvtrup, S. (1977). The Phylogeny of Vertebrata. Wiley, New York.
Maisey, J. G. (1986). Heads and tails: a chordate phylogeny. Cladistics, 2, 201-256.
Maisey, J. G. (1988). Phylogeny of Early vertebrate skeletal induction and ossification pattern. Evolutionary Biology, 22, 1-36.
Moy-Thomas, J. A. and Miles, R. S. (1971). Palaeozoic Fishes, 2nd edn, extensively revised by R. S. Miles. Chapman and Hall, London.
Nelson, G. J. (1969). Gill arches and the phylogeny of fishes, with notes on the classification of vertebrates. Bulletin of the American Museum of Natural History, 141, 475-552.
Nelson, G. J. (1989). Phylogeny of major fish groups. In The hierarchy of life (ed. B. Fernholm, K. Bremer, and H. Jörnvall), pp. 325-336, Excerpta Medica, Amsterdam.
Northcutt, R.G. and Gans, C. (1983). The genesis of neural crest and epidermal placodes: a reinterpretation of vertebrate origins. The Quarterly Review of Biology, 58, 1-28.
Schaeffer, B. and Thomson, K. S. (1980). Reflections on agnathan-gnathostome relationships. In Aspects of vertebrate life (ed. L. L. Jacobs), pp. 19-33. Museum of Northern Arizona Press, Flagstaff.
Yalden, D. W. (1985). Feeding mechanisms as evidence for cyclostome monophyly. Zoological Journal of the Linnean Society, 84, 291-300.
About This Page

Muséum National d'Histoire Naturelle Paris, France
Page copyright © 1997
 Page: Tree of Life
Craniata. Animals with skulls.
Authored by
Philippe Janvier.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Craniata. Animals with skulls.
Authored by
Philippe Janvier.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Citing this page:
Janvier, Philippe. 1997. Craniata. Animals with skulls. Version 01 January 1997 (under construction). http://tolweb.org/Craniata/14826/1997.01.01 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site