Isopoda
Richard Brusca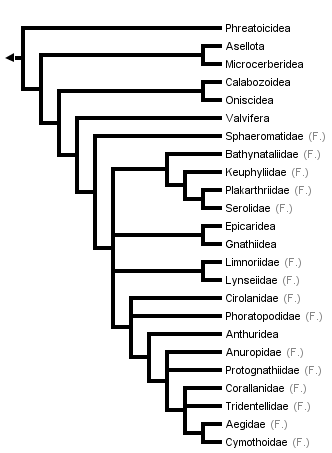


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxTree from Brusca & Wilson (1991).
Introduction
Isopods are the most diverse in form and the most species-rich crustaceans of the superorder Peracarida (isopods, amphipods, tanaids, and their kin). Isopods are common inhabitants of nearly all environments, and they are unusual among the Crustacea for their ubiquity. The familiar sowbugs and pillbugs are members of this group, along with their marine relatives (gribbles, slatters, etc.). The Isopoda include approximatly 10,000 described species, in 10 suborders. These animals range in length from 0.5 mm to 500 mm (Bathynomus giganteus). Phylogenetic analyses and the fossil record (limited though it is) suggest that the group dates to at least the Carboniferous Period of the Paleozoic, approximately 300 million years ago.
Characteristics
As in most crustaceans, the isopod body is divided into three distinct regions: head (= cephalon), thorax, and abdomen (= pleon). In isopods, the first segment of the thorax is fused to the head. The remaining seven free segments (pereonites) of the thorax comprise the pereon; each normally bears a pair of uniramous legs, or pereopods. The pereopods are modified for locomotion and for latching onto prey. In isopods, the abdomen primitively consists of 5 free segments (pleonites) plus a fused 6th pleonite+telson (the pleotelson). Each pleonite bears a pair of biramous pleopods, which are used for swimming and for respiration. Isopods have compound eyes, two pairs of antennae, and four sets of jaws. The first antennae are typically chemosensory; the second antennae are typically tactile structures. The jaws are (anterior to posterior): mandibles, maxillae 1, maxillae 2, maxillipeds. As the name implies, the maxillipeds are actually the highly modified appendages of the fused first thoracic segment.

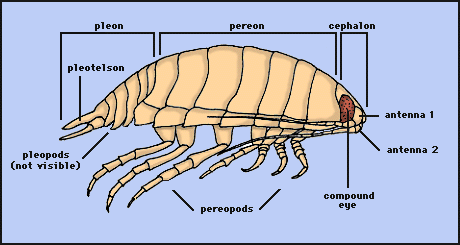
Drawing copyright © 1997 R. Brusca.
The Isopoda are a monophyletic clade defined by 13 synapomorphies (shared, derived characters):
- sessile eyes (i.e. not on a stalk)
- carapace reduced to a cephalic shield
- thoracopods entirely uniramous
- second antennae uniramous
- pleomere 6 fused to telson, forming a pleotelson
- biphasic molting
- heart thoraco-abdominal
- principal gas exchange structures are pleopods
- gut tube entirely ectodermally derived, without an endodermal midgut region
- striated muscles with unique myofibril ultrastructure
- loss of the maxillulary palp
- first antennae (antennules) uniramous, without a scale (scales secondarily reappear in the cirolanid genus Bathynomus, in the family Limnoriidae, and perhaps in the suborder Epicaridea)
- uropodal rami always uniarticulate.
Synapomorphies 1-4 appear to be convergent in isopods and amphipods; synapomorphy 5 may be convergent to the condition in many tanaidaceans (or may be a synapomorphy uniting isopods and tanaids).
Habitats
Approximately 4,500 marine species are known from all regions of the world's ocean, living most abundantly on the sea bottom from the abyss to the intertidal zone, and with only a few representatives in the pelagic zone. The group also has successfully colonized freshwater habitats (about 500 species) including lakes, rivers, streams, underground waters, thermal springs, the water held in certain tropical plants, and anchialine/cave habitats, where they often display associated specializations. All marine shorelines in the world are inhabited by species of the three very common families Idoteidae (Valvifera), Sphaeromatidae (Flabellifera), and Cirolanidae (Flabellifera). In offshore, soft-bottom, marine environments species of the suborders Asellota, Anthuridea, and Gnathiidea are very common. In the deep sea asellotans predominate almost to the complete exclusion of all other isopod taxa and, in fact, have undergone a massive evolutionary radiation in this environment (Wilson & Hessler 1987). Members of the suborder Anthuridea are slender, tubular forms that inhabit both shallow-water and shelf environments, and they are especially diverse in tropical reef habitats. Members of the suborder Oniscidea (about 5,000 species) are fully terrestrial (sowbugs and pillbugs), and they are by far the most successful group of crustaceans to invade land.
Feeding
Isopod feeding habits are extremely diverse. Within specific habitats, the isopods frequently constitute a major component of the energy cycle, fulfilling roles of micrograzers, micropredators, parasites, and detritivores. In general, the primitive suborders (e.g. Phreatoicidea, Asellota, Microcerberidea, Calabozoidea, Oniscidea, Valvifera) are herbivores or herbivorous scavengers, whereas the more derived suborders (e.g. Flabellifera, Epicaridea, Gnathiidea) are carnivores, predators, and parasites. Herbivores typically have grinding mandibles (with flat molars), whereas carnivores typically have cutting mandibles (with bladelike molars). When present in moderate to high numbers, herbivorous isopods probably have a significant impact on vegetation. For example, Perry & Brusca (1989) found that root-boring isopods significantly affect mangrove growth and prop root formation. Some groups are exclusively (suborder Epicaridea) or partly (suborder Flabellifera) parasitic. In some areas of the world, isopods emerge from the benthos in large numbers at night to prey on (and frequently kill) diseased or injured fishes, as well as attacking fishes caught in commercial traps or nets (Stepien & Brusca 1985).


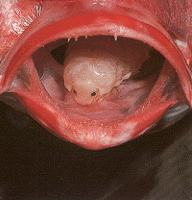
Left: Nerocila acuminata is a parasitic isopod that attaches to the skin of a variety of marine fishes. Image © Peter Wirtz. Right: The tongue-eating isopod, Cymothoa exigua, causes degeneration of the tongue of its host fish, the rose snapper, Lutjanus guttatus, and it then attaches to the remaining tongue stub and floor of the fish's mouth by hook-like pereopods. In this position the isopod superficially resembles its host's missing tongue. Brusca & Gilligan (1983) hypothesize that these isopods serve as a mechanical replacement for the fish's tongue and represent the first known case in animals of functional replacement of a host structure by a parasite. This relationship is so-far known only from the Gulf of California. © 1989 Matthew Gilligan, Savannah State College, Savannah, GA
Development
As with all the Peracarid crustaceans, isopod embryos undergo direct development within the female brood pouch (marsupium), from which they emerge as juveniles, known as manca. These manca are essentially replicas of adults but lack the last pair of thoracic legs. There is no pelagic larval stage among the isopods. Dispersal appears to be limited to the crawling ability of these forms, and this attribute has resulted in most species having highly restricted distributions and patterns of high endemism.
Discussion of Phylogenetic Relationships
The only parsimony-based explicit phylogenetic analysis of higher-level isopod phylogeny was that of Brusca & Wilson (1991, data matrix available from TreeBase). While they concluded that the Isopoda comprise a monophyletic group (a true evolutionary group descending from a single ancestor), they found that one of the ten suborders of isopods, Flabellifera, was not monophyletic. The families of this suborder are therefore presented separately in the phylogeny shown above. The paraphyly of the Flabellifera is clearly evidenced by the scattered locations of the families within the tree. This is an example of a currently used classification that does not accurately reflect the estimated phylogeny of the group in question.
The phylogeny of Brusca and Wilson shows the isopods originating in a "short-tailed" morphology similar to that of modern tanaids, in which the pleotelson is highly reduced and the styliform uropods and anus are terminal. The derived "long-tailed morphology," with a broad elongate pleotelson, flat laterally-placed uropods, and subterminal anus, distinguishes a transition from sedentary and infaunal lifestyles (typical of the short-tailed taxa) to a more active lifestyle typical of the advanced orders and families.
Biogeographical history
Isopods apparently evolved in shallow marine environments by at least the early or mid-Paleozoic. The first isopods were "short-tailed" - with short pleotelsons and terminal styliform uropods. Phylogenetic analyses and the fossil record agree that the earliest isopods (and the most primitive living species) are members of the short-tailed suborder Phreatoicidea. Today, phreatoicids have a strictly freshwater Gondwanan distribution, with most species occurring in the rivers and lakes of Tasmania. The earliest fossil records of isopods are phreatoicids dating from the Pennsylvanian (the Carboniferous Period of the Paleozoic Era), 300 million year ago. However, Paleozoic phreatoicids were marine forms and they had a cosmopolitan distribution; their fossils have been found in marine deposits from Europe and North America. Thus, the present-day Gondwanan freshwater distribution of these primitive crustaceans represents a relic, or refugial biogeographic pattern.
In fact, virtually all of the short-tailed isopod taxa occupy what could be regarded as biogeographical refugia. Primitive Asellota are largely fresh-water or ground-water inhabitants. Higher Asellota live primarily in the deep-sea, an environment nearly uninhabited altogether by other isopod taxa. Microcerberidea inhabit coastal ground waters or are interstitial. Calabozoidea are so far known only from freshwater springs in Venezuela. And Oniscidea have escaped the aquatic world altogether and are the only fully terrestrial crustaceans. In all these cases, it seems that the primitive isopod lineages have found environments that allowed them to escape the challenges of predation by shallow-water marine fishes, their principal predators, which began their major radiation at about the same time the isopods were beginning theirs (in the middle Paleozoic). It may be that the evolution and radiation of the more advanced "long-tailed" isopods, which probably began in the late Triassic (Mesozoic), also helped drive the short-tailed forms into refugia by competition. Unlike short-tailed taxa, species of long-tailed isopods are highly mobile and frequently leave their benthic shelter to swim short distances through the water. Their elongate pleotelsons and broad, laterally-positioned uropods provide hydrodynamic planar surfaces to assist in swimming.
The profound shift from a relatively sedentary, short-tailed morphology to the active, long-tailed form appears to have coincided with the fragmentation of Pangaea. Since the majority of the long-tailed higher taxa are endemic to the Southern Hemisphere, this suggests that they originated on Gondwanan shores shortly after its separation from Laurasia.
Studies of isopod biogeography have been largely restricted to two suborders, the Valvifera and the Asellota. Among the suborder Valvifera, the family Idoteidae is thought to have co-evolved with large brown algae (Phaeophyta; Laminariales) in temperate latitudes (Brusca and Wallerstein 1979). Idoteids are primarily temperate in distribution and closely associated with the cold-water seaweeds upon which they feed and live. The southern range limits of temperate idoteid isopods may be controlled today primarily by biotic factors, such as absence of suitable substrate/food (e.g. laminarians) or predation pressure from tropical shallow-water fishes. Wallerstein and Brusca (1982) documented patterns of idoteid morphology and behavior indicating that when these isopods evolve in warmer waters they are smaller, their bodies are more spinose, their swimming behaviors differ from their temperate cousins, and they also reproduce at smaller sizes; all possible adaptations to avoid increased predation pressure in the tropics. A phylogenetic analysis of the Valvifera suggested that the suborder originated in the temperate Southern Hemisphere, at least by Permian/Triassic times (Brusca 1984); global distribution patterns of some genera can be ascribed to vicariance processes, others to dispersal, ecological phenomena, or a combination of processes.
Of the 10 isopod suborders, 4 have representatives in the deep sea. However, one of these 4 suborders, the Asellota is far and away the predominant deep-sea isopod taxon, and about 90 percent of all described isopods from this environment are asellotans. Wilson (1980) and Wilson and Hessler (1987) argued that the deep sea has been invaded numerous times by asellotans of several families. Here, the Asellota have radiated widely. In polar regions, various asellotan lineages have re-emerged into shallower waters.
Hessler and Wilson (1983) calculated that 32-51% of all benthic species taken in all deep-sea samples, anywhere in the world have been peracarids, and of the peracarids the isopods are by far the most abundant and speciose deep-sea group. Research has shown that most deep-sea genera are cosmopolitan, whereas deep-sea species may be either widespread or highly restricted in their distribution.
References
Brusca, R. C. 1984. Phylogeny, evolution and biogeography of the marine isopod subfamily Idoteinae (Crustacea: Isopoda: Idoteidae). Trans. San Diego Soc. Nat. Hist. 20: 99-134.
Brusca, R. C. and M. R. Gilligan. 1983. Tongue replacement in a fish by a parasitic Isopod. Copeia. 1983(3):813-816.
Brusca, R. C. and E. W. Iverson. 1985. A Guide to the Marine Isopod Crustacea of Pacific Costa Rica. Revista de Biologia Tropical 33, Supplement 1:1-77.
Brusca, R. C. and B. R. Wallerstein. 1979. Zoogeographic patterns of idoteid isopods in the northeast Pacific, with a review of shallow-water zoogeography for the region. Bull. Biol. Soc. Wash. 3: 67-105.
Brusca, R. C. and G. D. F. Wilson. 1991. A phylogenetic analysis of the Isopoda with some classificatory recommendations. Mem. Queensland Mus. 31: 143-204.
Brusca, R. C., R. Wetzer, & S. France. 1995. Cirolanidae (Crustacea: Isopoda: Flabellifera) of the Tropical Eastern Pacific. Proc. San Diego Soc. Nat. Hist. 30: 1-96.
Cohen, B. J. & G. C. B. Poore. 1994. Phylogeny and biogeography of the Gnathiidea (Crustacea: Isopoda) with descriptions of new genera and species most from south-eastern Australia. Mem. Mus. Victoria 54: 271-397.
Delaney, P. M. 1989. Phylogeny and biogeography of the marine isopod family Corallanidae (Crustacea, Isopoda, Flabellifera). Contrb. Sci., L.A. Co. Nat. Hist. Mus. 409: 1-75.
Grassle, J. F. & N. J. Maciolek. 1992. Deep-sea species richness: Regional and local diversity estimates from quantitative bottom samples. Am. Nat. 139:313-341.
Hessler, R.R. and G.D.F. Wilson. 1983. The origen and biogeography of malacostracan crustaceans in the deep sea. in, R.W. Sims, J.H. Price and P.E.S. Whalley, 1983, Evolution, Time and Space: The Emergence of the Biosphere, Academic Press, N.Y. [Systematics Assoc. Special Vol. No. 23]. pp. 227-254.
Hopkin, S. P. 1989. Ecophysiology of Metals in Terrestrial Invertebrates. Elsevier Applied Science, Barking.
Kensley, B. 1984. The role of isopod crustaceans in the reef crest community at Carrie Bow Cay, Belize. Mar. Ecol. 5(1):29-44.
Kensley, B. and R. C. Brusca (eds.) 2001. Isopod Systematics and Evolution. Balkema, Rotterdam.
Kensley, B. & M. Schotte. 1989. Guide to the Marine Isopod Crustaceans of the Caribbean. Smithsonian Institution Press, Washington, D.C.
Kensley, B. In Press. Estimates of species diversity of free-living marine isopod crustaceans of coral reefs. Coral Reefs.
Kensley, B., S. Schilling, & M. Schotte. 1996. World List of Terrestrial Isopods (Oniscidea). Smithsonian Institution Gopher Server, accessible via Smithsonian Home Page on WWW. In draft version.
Kensley, B. & M. Schotte. 1995. World List of Marine and Freshwater Isopods. Smithsonian Institution Gopher Server, accessible via Smithsonian Home Page on WWW.
Moore, W. ms. Historical biogeography of the central tropical Pacific based on anthuridean isopod crustaceans. In draft version.
Perry, D. M. and R. C. Brusca. 1989. Effects of the root-boring isopod Sphaeroma peruvianum on red mangrove forests. Mar. Ecol. Prog. Ser. 57: 287-292.
Poore, G. C. B. & G. Wilson. 1993. Marine species richness. Nature, London 361: 587-583.
Richardson, H.R. 1905. A Monograph on the Isopods of North America. Bulletin of the United States National Museum 54:1-727.
Salemaa, H. 1978. Geographic variability in the colour polymorphism of Idotea baltica (Isopoda) in the northern Baltic. Hereditas 88: 165-182.
Salemaa, H. 1979. Seasonal variability in the colour polymorphism of Idotea baltica (Isopoda) in the northern Baltic. Hereditas 90: 51-57.
Schram, F. R. 1970. Isopods from the Pennsylvanian of Illinois. Science 169: 854-855.
Schram, F. R. 1974. Paleozoic Peracarida of North America. Fieldiana, Geology 33: 95-124.
Stepien, C. A. & R. C. Brusca. 1985. Nocturnal attacks on nearshore fishes in southern California by crustacean zooplankton. Mar. Ecol. Prog. Ser. 25: 91-105.
Sutton, S. 1972. Woodlice. Ginn & Co., London. 144 pages.
Vandel, A. 1960. Isopodes Terrestres (Premiere Partie). Faune de France 64:1-41 6.
Wallerstein, B. R. & R. C. Brusca. 1982. Fish predation: A preliminary study of its role in the zoogeography and evolution of shallow-water idoteid isopods (Crustacea: Isopoda: Idoteidae). J. Biogeography 9: 135-150.
Wilson, G. D. F. 1980. Superfamilies af the Asselota (Isopoda) and the systematic position of Stenetrium weddellense (Schultz). Crustaceana, 38(2): 219-221.
Wilson, G. D. F. and R. R. Hessler. 1987. Speciation in the deep sea. An. Rev. Ecol. Syst. 18: 185-207.
Information on the Internet
- The Smithsonian's World Isopod List includes a listing of all marine and terrestrial isopod species as well as a bibliography of the species descriptions.
- The Museum of Victoria's page on the Isopods of Southern Australia.
- Page on the parasitic epicaridean isopods.
Title Illustrations

| Scientific Name | Sphaeromene polytylotos |
|---|---|
| Comments | The button isopod is found amongst mussels and barnacles on rocky shores and reefs along the coast of western South Africa. It is herbivorous, feeding on filamentous algae and on simple green algae. |
| Reference | G. M. Branch, C. L. Griffiths, M. L. Branch, and L. E. Beckley. 1994. Two Oceans, a Guide to the Marine Life of Southern Africa. David Philip Publishers, South Africa. |
| Specimen Condition | Live Specimen |
| Copyright | © 1994 G. M. Branch |
| Scientific Name | Armadillidium vulgare |
|---|---|
| Location | Catania |
| Specimen Condition | Live Specimen |
| Identified By | Giuseppe Montesanto |
| Sex | Male |
| Copyright |
© 2006

|
| Scientific Name | Deto echinata |
|---|---|
| Comments | As the name suggests, the horned isopod is easily recognized by the pairs of "horns" arising on the back of each thoracic segment. It is often confused with the similar looking Ligia, to which it is only distantly related. It is an air breathing isopod found associated with kelp and other drift algae washed up on rocky shores of western South Africa. It feeds mainly on this algae as well as live prey and carrion. |
| Reference | G. M. Branch, C. L. Griffiths, M. L. Branch, and L. E. Beckley. 1994. Two Oceans, a Guide to the Marine Life of Southern Africa. David Philip Publishers, South Africa. |
| Specimen Condition | Live Specimen |
| Copyright | © 1994 G. M. Branch |
About This Page
This Tree of Life page was composed and assembled primarily by Sarah Riseman, while she was a MSc student and National Science Foundation PEET trainee at the University of Charleston.
Richard Brusca

Arizona-Sonora Desert Museum, Tucson, Arizona, USA
Page copyright © 1997 R. Brusca
 Page: Tree of Life
Isopoda.
Authored by
Richard Brusca.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Isopoda.
Authored by
Richard Brusca.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 06 August 1997
Citing this page:
Brusca, Richard. 1997. Isopoda. Version 06 August 1997. http://tolweb.org/Isopoda/6320/1997.08.06 in The Tree of Life Web Project, http://tolweb.org/












 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site