Latrodectus
Widow spiders
Jeremy Miller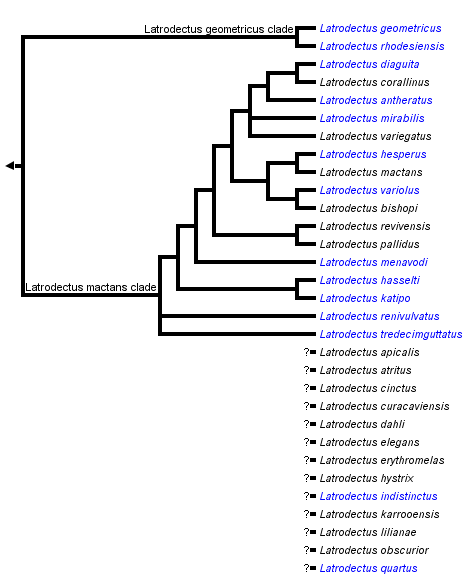


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Widow spiders (genus Latrodectus) are widely feared but poorly known. They are a medically important group with a worldwide distribution. Most species favor xeric conditions. Some species adapt well to synanthropic (human-altered) conditions and are readily dispersed by humans (Forster, 1984; 1985; Forster and Forster, 1999; Garb et al., 2004; Ono, 1995; Ori et al., 1996). They have remarkably diverse sexual biology involving cannibalism, kleptoparasitism, mate guarding, mating plugs, post-copulatory sterility, and varying degrees of sexual size dimorphism (e.g., Anava and Lubin, 1993; Andrade, 1996; 1998; Andrade and Banta, 2002; Breene and Sweet, 1985; Forster, 1992; 1995; Ross and Smith, 1979; Segev et al., 2003; Snow et al., 2005; Snow and Andrade, 2005a; b).
There are currently 31 valid species of Latrodectus. Widow spiders are most diverse in Argentina (seven species), the Middle East (seven species), South Africa (six species), and North America (five species). There are undescribed species and nomenclatural problems, so the taxonomy of the group is unstable.
Characteristics
The male palp (intromittent organ) is distinctive with a long spiral embolus. The epigynum (female genitalia) features an atrium (large unified copulatory opening leading to a pair of independent copulatory ducts) and dumbbell-shaped spermathecae (female sperm storage organs). The famous red hourglass marking on the ventral part of the abdomen is not present in all species.
Medical Importance
Black widow venom is a neurotoxin that can cause latrodectism, a mild to severe medical syndrome. The bite itself is usually not perceived but local pain may occur several minutes after the bite. Typical symptoms of latrodectism include swelling of the lymphatic nodes, profuse sweating, rigidity of the abdominal muscles, facial contortions, and hypertension (Maretic, 1971; 1975; 1983). Antivenin is available to counteract the effects of Latrodectus. Cases of latrodectism are known from all geographic regions where widow spiders occur. Across the globe envenomations appear to be patchy. Actual or alleged envenomations in the various geographic regions are attributed to the various local widow species, which differ significantly in toxicity, aggressiveness, and ecology (Finlayson, 1956; Maretic, 1971; 1978; 1983; 1987; McCrone, 1964; McCrone and Netzloff, 1965; Müller et al., 1989; Müller et al., 1992; Shulov, 1940; Shulov and Weissman, 1959). Even without antivenin treatment, the mortality rate is low (on the order of 5%, mostly in small children; Schmidt, 1993).
One of the key factors influencing the epidemiology of latrodectism is the ecology of local Latrodectus species. Latrodectus tends to be urbanized in North America and Australia, while in Argentina and the Mediterranean region, Latrodectus is more likely to be found in rural or natural areas (Maretic, 1971; 1978). In regions where Latrodectus is not urbanized, latrodectism is often associated with farm workers and summer months, when farming is most intensive. In regions with urbanized Latrodectus species, latrodectism affects a broader cross section of the population, and bites may occur at almost any time of the year (Maretic, 1978).
Discussion of Phylogenetic Relationships
Garb et al. (2004) have produced the first phylogeny of Latrodectus species using the mitochondrial gene cytochrome oxidase I. This analysis produced a generally well supported tree. Species not included in the Garb et al. study are listed below the tree on this page.
References
Abalos, J. W. 1962. The egg-sac in the identification of species of Latrodectus (black-widow spiders). Psyche 69:268-270.
Abalos, J. W. 1980. Las arañas del género Latrodectus en la Argentina. Obra del Centenario del Museo de La Plata 6:29-51.
Abalos, J. W., and E. C. Báez. 1967. The spider genus Latrodectus in Santiago del Estero, Argentina. Pages 59-74 in Animal Toxins Pergamon Press, Oxford.
Anava, A., and Y. Lubin. 1993. Presence of gender cues in the web of a widow spider, Latrodectus revivensis, and a description of courtship behavior. Bull. Br. arachnol. Soc. 9:119-122.
Andrade, M. C. B. 1996. Sexual selection for male sacrifice in the Australian redback spiders. Science 271:70-72.
Andrade, M. C. B. 1998. Female hunger can explain variation in cannibalistic behavior despite male sacrifice in redback spiders. Behav. Ecol. 9:33-42.
Andrade, M. C. B., and E. M. Banta. 2002. Value of male remating and functional sterility in redback spiders. Animal Behav. 63:857-870.
Breene, R. G., and M. H. Sweet. 1985. Evidence of insemination of multiple females by the male black widow spider, Latrodectus mactans (Araneae, Theridiidae). J. Arachnol. 13:331-335.
Finlayson, M. H. 1956. "Knopie-Spider" bite in Southern Africa. Med. Proc. 2:634-638.
Forster, L. M. 1984. The Australian redback spider (Latrodectus hasselti): its introduction and potential for establishment and distribution in New Zealand. Pages 273-289 in Commerce and the Spread of Harmful Pests and Disease Vectors (M. Laird, ed.) New York, Praeger Press.
Forster, L. M. 1985. Is the redback spider here to stay? New Zealand J Ag. 150:58-59.
Forster, L. M. 1992. The stereotyped behavior of sexual cannibalism in Latrodectus hasselti Thorell (Araneae: Theridiidae), the Australian redback spider. Aust. J. Zool. 40:1-11.
Forster, L. M. 1995. The behavioral ecology of Latrodectus hasselti (Thorell), the Australian redback spider (Araneae: Theridiidae): A review. Rec.Western Australian Mus., Sup 52:13-24.
Forster, R., and L. Forster. 1999. Spiders of New Zealand and their World-Wide Kin. University of Otago Press, Hong Kong.
Garb, J. E., A. González, and R. G. Gillespie. 2004. The black widow spider genus Latrodectus (Araneae: Theridiidae): phylogeny and invasion history. Mol. Phy. Evol. 31:1127-1142.
Knoflach, B., and A. van Harten. 2002. The genus Latrodectus (Araneae: Theridiidae) from mainland Yemen, the Socotra Archipelago and adjacent countries. Fauna Arabia 19:321-361.
Levi, H. W. 1966. The three species of Latrodectus found in Israel. J. Zool., London 150:427-432.
Levi, H. W. 1983. On the value of genitalic structures and coloration in separating species of widow spiders (Latrodectus sp.) (Arachnida: Araneae: Theridiidae). Verh. Naturwiss. Ver. Hamburg 26:195-200.
Levy, G., and P. Amitai. 1983. Revision of the widow-spider genus Latrodectus (Araneae: Theridiidae) in Israel. Zool. J. Linnean Soc. 77:39-63.
Lotz, L. N. 1994. Revision of the genus Latrodectus (Araneae: Theridiidae) in Africa. Navorsinge van die Nasionale Museum Bloemfontein 10:1-60.
Maretic, Z. 1971. Latrodectism in Mediterranean countries, including South Russia, Israel, and North Africa. Pages 299-309 in Venomous Animals and their Venoms. Volume III. Venomous Invertebrates (W. Bücherl, and E. E. Buckley, eds.). Academic Press, London.
Maretic, Z. 1975. European araneism. Bull. Br. arachnol. Soc. 3:126-130.
Maretic, Z. 1978. Venoms of Theridiidae, genus Latrodectus. B. Epidemiology of envenomation, symptomatology, pathology and treatment. Pages 185-207 in Arthropod Venoms. Handbuch der experimentellen Pharmakologie. Volume 48 (S. Bettini, ed.) Springer Verlan, Berlin.
Maretic, Z. 1983. Latrodectism: Variations in clinical manifestations provoked by Latrodectus species of spiders. Toxicon 21:457-466.
Maretic, Z. 1987. Spider venoms and their effect. Pages 142-159 in Ecophysiology of Spiders (W. Nentwig, ed.) Springer Verlag, Berlin.
McCrone, J. D. 1964. Comparative lethality of several Latrodectus venoms. Toxicon 2:201-203.
McCrone, J. D., and M. L. Netzloff. 1965. An immunological and electrophoretical comparison of the venoms of the North American Latrodectus spiders. Toxicon 3:107-110.
Melic, A. 2000. El género Latrodectus Walckenaer, 1805 en la Península Ibérica (Araneae: Theridiidae). Rev. Iber. Aracnol. 1:13-30.
Müller, G. J., H. M. Koch, A. B. Kreigler, B. J. van del Walt, and P. P. van Jaarsveld. 1989. The relative toxicity and polypeptide composition of the venom of two Southern African widow spider species: Latrodectus indistinctus and Latrodectus geometricus. South African J. Sci. 85:44-46.
Müller, G. J., A. B. Kreigler, J. M. van Zyl, B. J. van del Walt, A. S. Dippenaar, and P. P. van Jaarsveld. 1992. Comparison of the toxicity, neurotransmitter releasing potency and polypeptide composition of the venoms from Steatoda foravae, Latrodectus indistinctus and L. geometricus. South African J. Sci. 88:113-116.
Ono, H. 1995. Records of Latrodectus geometricus (Araneae: Theridiidae) from Japan. Acta arachnol. 44:167-170.
Ori, M., E.-I. Shinkai, and H. Ikeda. 1996. Introduction of widow spiders into Japan. (In Japanese). Med. Ent. Zool. 47:111-119.
Ross, K., and R. L. Smith. 1979. Aspects of the courtship behavior of the black widow spider, Latrodectus hesperus (Araneae: Theridiidae), with evidence for the existence of a contact sex pheromone. J. Arachnol. 7:69-77.
Schmidt, G. 1993. Giftige und gefährliche Spinnentiere. Westarp Wissenschaften, Magdeburg, Essen.
Segev, O., M. Ziv, and Y. Lubin. 2003. The male mating system in a desert widow spider. J. Arachnol. 31:379-393.
Shulov, A. 1940. On the biology of two Latrodectus spiders in Palestine. Proc. Linn. Soc., London 152:309-328.
Shulov, A., and A. Weissman. 1959. Notes on the life habits and potency of the venom of the three Latrodectus spider species of Israel. Ecology 40:515-518.
Snow, L. S. E., A. Abdel-Mesih, and M. C. B. Andrade. 2005. Broken copulatory organs are low-cost adaptations to sperm competition in redback spiders. Ethology 111:1-17.
Snow, L. S. E., and M. C. B. Andrade. 2005a. Multiple sperm storage organs facilitate female control of paternity. Proc. R. Soc. Lond. B 272:1139-1144.
Snow, L. S. E., and M. C. B. Andrade. 2005b. Pattern of sperm transfer in redback spiders: implications for sperm competition and male sacrifice. Behav. Ecol. 15:785-792.
Title Illustrations

| Scientific Name | Latrodectus hesperus |
|---|---|
| Location | Chatfield Reservoir, Douglas Co., Colorado |
| Specimen Condition | Live Specimen |
| Identified By | Jeremy Miller |
| Sex | Female |
| Life Cycle Stage | Adult |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
©

|
About This Page

Nationaal Natuurhistorisch Museum Naturalis, Leiden, The Netherlands
Correspondence regarding this page should be directed to Jeremy Miller at
Page copyright © 2006
All Rights Reserved.
- First online 08 January 2007
- Content changed 08 January 2007
Citing this page:
Miller, Jeremy. 2007. Latrodectus. Widow spiders. Version 08 January 2007 (under construction). http://tolweb.org/Latrodectus/93274/2007.01.08 in The Tree of Life Web Project, http://tolweb.org/







 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site