Linepithema
Alex Wild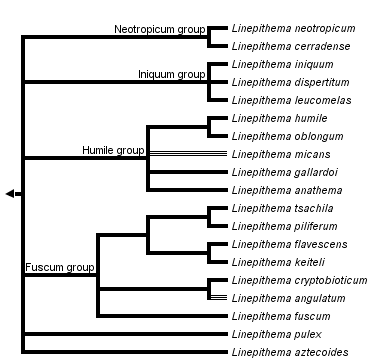


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction


Linepithema micans workers, Argentina © Alex Wild
Linepithema is a lineage of small, monomorphic dolichoderine ants native from Mexico and the Caribbean south to Argentina. The group contains 19 described species (Wild 2008) but is widely recognized for a single pest species, the Argentine ant L. humile (Mayr) that is ecologically invasive in warmer climates worldwide (Roura-Pasqual et al 2004, Wetterer et al 2009). The Argentine ant is listed as one of the 100 worst invasive species worldwide by the IUCN and has received extensive study as a model species for invasion biology (Holway et al 2003, Tsutsui et al 2001, Giraud et al 2001).
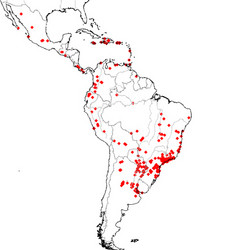
The native distribution of Linepithema based on museum specimens. Records of introduced L. humile are excluded.
The natural history of most species is poorly known. Where documented, Linepithema ants live in large colonies of at least 1,000 workers, and often many more, with some populations forming sprawling "supercolonies" covering tens to thousands of meters (Sunamura et al 2009, Tsutsui and Case 2001). Most species nest in soil or leaf litter, but L. iniquum and L. leucomelas are primarily arboreal.
Linepithema is a trophic generalist. Ants gather protein from a wide variety of scavenged or hunted sources, and like most other dolichoderines they consume significant amounts carbohydrates in the form of insect honeydew both inside and outside the nest (Wild 2008). Some species keep root aphids in subterranean galleries. Workers recruit to food sources in great number using persistent pheromone trails, and similar trails are often maintained among nest sites (Wild 2008).
Linepithema colonies are highly variable in structure both within and among species (Wild 2008). They may have a single queen (monogyny) to hundreds of queens (polygyny), inhabit a single nest (monodomy) or many nests (polydomy), and reproduce either by independently dispersing queens or by colony fission where queens and workers found new colonies as a group (Wild 2008). The tendency of L. humile in particular to form massive multi-nest, multi-queen colonies that reproduce through fission is thought to be a factor in its conversion to an invasive pest (Tsutsui and Case 2001).
Worker ants in a colony are monomorphic, i.e., of similar size and body proportions, and can be monotonously similar across species, rendering species-level identifications from worker specimens difficult (Wild 2004, Wild 2008). In contrast, Linepithema males show striking differences among species in overall size and body proportions as well as in wing venation and genital morphology (Wild 2008). The reasons behind the diversity of male forms is unknown, but may be related to divergent colony-founding strategies involving flighted and flightless queens.


A selection of habitats where Linepithema is native. A, montane pine forest in Guatemala
(L. dispertitum); B, riparian palm forests in Argentina (L. humile, L. micans); C, humid
subtropical tall forest in Paraguay (L. pulex, L. neotropicum, L. iniquum); D, high Andean
grasslands in Argentina (L. oblongum).
Characteristics

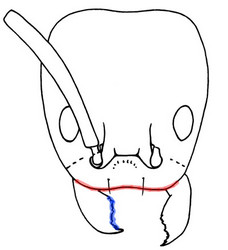
Head in full-face view of Linepithema keiteli
from the Dominican Republic. Diagnostic features
for Linepithema include the medially concave anterior
clypeal margin (in red) and the series of alternating
teeth and denticles on the mandible (in blue).
Female Linepithema (both queens and workers) can be uniquely identified by the following combination of character states, also illustrated in the above figure:
1. Anteromedial clypeal margin with a broad, shallow concavity.
2. Masticatory margin of the mandibles exclusive of apical and subapical teeth formed by a series of 3-4 teeth alternating with smaller denticles.
Additional characters that may help separate Linepithema workers from related genera are:
3. Compound eyes with 15-110 ommatidia, centered anterior of midline in full-face view, and not touching lateral margins.
4. Maxilla bearing 6 palpomeres and labia with 4 palpomeres.
5. Mesosoma lacking spines or teeth.
6. Propodeum in lateral view depressed below level of mesonotum (only slightly in L. cryptobioticum).
7. Abdominal sternite 6 (= gastric sternite 4) keel-shaped posteriorly.
Male Linepithema (excepting rare worker-like forms in L.dispertitum) can be separated from males of related genera by the following combination of character states:
1. Antennal scape shorter than third antennal segment.
2. Lateral ocelli emerging above posterior cephalic margin in full face view.
3. Anteromedial clypeal margin broadly convex.
4. Mandibles with a distinct masticatory margin bearing at least 4 teeth or denticles, sometimes approaching worker dentition.
5. Mesoscutum covered with dense, fine pubescence.
6. Petiolar scale not inclined anteriorly, instead with scale straight, inclined posteriorly, or present as a low node.
7. Forewings with 1–2 closed submarginal cells.
8. Volsella with digitus narrow and sharply downturned distally.


The two male forewing types in Linepithema, illustrating the derived Fuscum-group
venation with two submarginal cells (top) and the general ancestral condition (bottom) found
elsewhere in the genus. The submarginal cells are colored green for easy visualization.
Discussion of Phylogenetic Relationships
The monophyly of Linepithema is well-established. The group is supported by numerous morphological characters, including features of the female clypeus and male genitalia (Wild 2008, Shattuck 1992b), and by multi-locus molecular studies of Wild (2009) and Ward et al (2010). Ward et al (2010) estimated the age of the divergence of Linepithema from its sister clade as occurring between 24 and 45 mya and the age of divergence of the extant species between 4 and 13 mya.
Martins (2007) produced the first species-level phylogeny based on mitochondrial sequences from several Brazilian samples, recovering a clade comprising L. humile, L. micans, and L. gallardoi. Wild (2009) followed with a larger matrix employing genetic data from four loci (COI, ITS-2, Long-wavelength rhodopsin, and wingless) and 45 specimens from across the generic range. The result is summarized at the top of this page. Four deeper clades consistently emerged in the analyses (Wild 2009). These were broadly congruent with morphological characters (Wild 2008) and were used to establish species groups as follows:
| species group | male forewing, submarginal cell count | male propodeal profile | volsellar cuspis | worker metapleural pubescence | worker propodeal profile |
|---|---|---|---|---|---|
| Fuscum | 2 | convex | absent | sparse | variable |
| Humile | 1 | strongly concave | present | dense | inclined forward above spiracle |
| Iniquum | 1 | convex | present | sparse | variable |
| Neotropicum | 1 | weakly concave | present | sparse to moderate | rounded, with dorsal longer the posterior face |
Two south American species, L. pulex and L. aztecoides, emerged basally without clear association with the species groups (Wild 2009).


Linepithema aztecoides, Paraguay © Alex Wild
References
Bolton, B. 1994. Identification Guide to the Ant Genera of the World. 222 pp. Cambridge, Mass.
Bolton, B., G. Alpert, P. S. Ward, and P. Naskrecki. 2007. Boltons Catalogue of ants of the world. Cambridge, MA: Harvard University Press, CD-ROM.
Bolton, B. 2003. Synopsis and Classification of Formicidae. 370 pp. Memoirs of the American Entomological Institute, Vol. 71. Gainesville, FL.
Brandão, C. R. F., C. Baroni Urbani, J. Wagensberg, and C. I. Yamamoto. 1998. New Technomyrmex in Dominican amber, with a reappraisal of Dolichoderinae phylogeny. Entomologica Scandinavica 29: 411-428.
Giraud, T., J. S. Pedersen, and L. Keller. 2002. Evolution of supercolonies: The Argentine ants of southern Europe. Proc. Natl. Acad. Sci. USA 99: 6075-6079.
Hölldobler, B. and E. O. Wilson. 1990. The Ants. 732 pp. Harvard University Press.
Holway, D. A., L. Lach, A. V. Suarez, N. D. Tsutsui, and T. J. Case. 2002. The Causes and Consequences of Ant Invasions, Annual Review of Ecology and Systematics 33: 181-233.
Martins, C. 2007. Ferramentas moleculares parao estudode formigas: o caso do genero Linepithema (Formicidae: Dolichoderinae) do estado de Sao Paulo. Biologico, Sao Paulo 69(Suppl. 2): 6164.
Mayr, G. 1866. Myrmecologische Beiträge. Sitzungsber. Kais. Akad. Wiss. Wien Math.-Naturwiss. Cl. Abt. I 53: 484-517.
Mayr, G. 1868. Formicidae novae Americanae collectae a Prof. P. de Strobel. Annu. Soc. Nat. Mat. Modena 3: 161-178.
Roura-Pascual, N, A. V. Suarez, C. Gomez, P. Pons, Y. Touyama, A. L. Wild, A. T. Peterson. 2004. Geographic potential of Argentine ants (Linepithema humile Mayr) in the face of global climate change. Proceedings of the Royal Society of London, Series B 271: 2527-2534.
Shattuck, S. O. 1992a. Higher classification of the ant subfamilies Aneuretinae, Dolichoderinae and Formicidae. Systematic Entomology 17: 199-206.
Shattuck, S. O. 1992b. Generic revision of the ant subfamily Dolichoderinae (Hymenoptera: Formicidae). Sociobiology 21: 1-181.
Shattuck, S. O. 1994. Taxonomic catalog of the ant subfamilies Aneuretinae and Dolichoderinae. University of California Publications in Entomology 112: 1-241.
Shattuck, S. O. 1995. Generic-level relationships within the ant subfamily Dolichoderinae (Hymneoptera: Formicidae). Systematic Entomology 20: 217-228.
Sunamura, E., X. Espadaler, H. Sakamoto, S. Suzuki, M. Terayama and S. Tatsuki. 2009. Intercontinental union of Argentine ants: behavioral relationships among introduced populations in Europe, North America, and Asia. Insectes Sociaux 56: 143-147.
Tsutsui, N. D., and T. J. Case. 2001. Population genetics and colony structure in the Argentine ant (Linepithema humile) in its native and introduced ranges. Evolution 55: 976-985.
Ward, P. S., S. G. Brady, B. L. Fisher, and T. R. Schultz. 2010. Phylogeny and Biogeography of Dolichoderine Ants: Effects of Data Partitioning and Relict Taxa on Historical Inference. Systematic Biology 59: 342-362.
Wetterer, J. K., A. L. Wild, A. V. Suarez, N. Roura-Pascual, and X. Espadaler. 2009. Worldwide spread of the Argentine ant, Linepithema humile (Hymenoptera: Formicidae) Myrmecological News 12: 187-194.
Wild, A. L. 2008. Taxonomic revision of the ant genus Linepithema (Hymenoptera: Formicidae). University of California Press, 126: 1-151.
Wild, A. L. 2009. Evolution of the Neotropical Ant Genus Linepithema. Systematic Entomology 34:49-62.
Title Illustrations

| Scientific Name | Linepithema humile |
|---|---|
| Specimen Condition | Live Specimen |
| Identified By | Alex Wild |
| Sex | worker |
| Life Cycle Stage | adult |
| View | latural |
| Size | 3mm |
| Copyright |
© Alex Wild

|
About This Page
Alex Wild

University of Illinois, Urbana, Illinois, USA
Correspondence regarding this page should be directed to Alex Wild at
Page copyright © 2010 Alex Wild
All Rights Reserved.
- First online 06 September 2004
- Content changed 23 September 2010
Citing this page:
Wild, Alex. 2010. Linepithema. Version 23 September 2010. http://tolweb.org/Linepithema/22286/2010.09.23 in The Tree of Life Web Project, http://tolweb.org/








 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site