Neobatrachia
"Higher" Frogs
David Cannatella, Linda Ford, and Lori Bockstanz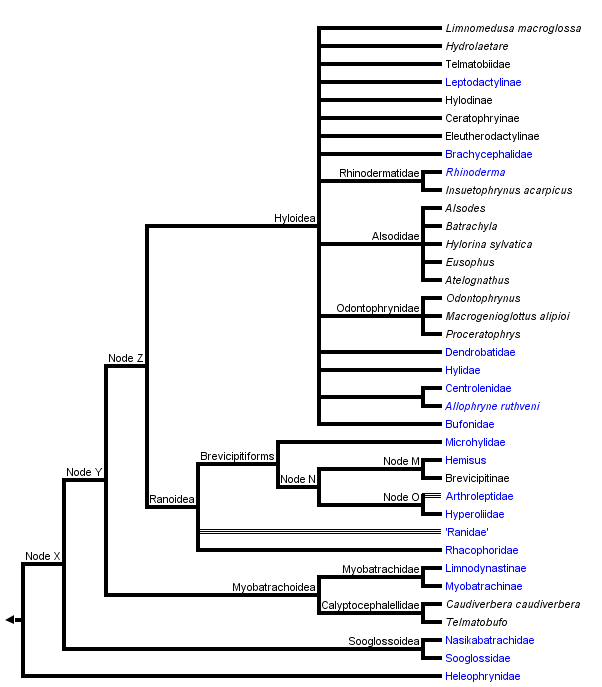


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Neobatrachians are sometimes called the "advanced" frogs. This group includes 95% of the species of frogs, but relationships among the major lineages are not well-resolved for the most part.
Discussion of Phylogenetic Relationships
Ford and Cannatella (1993) applied the node-based name Neobatrachia to the most recent common ancestor of living hyloids (myobatrachids, leptodactylids, bufonids, hylids, centrolenids, pseudids, sooglossids, Heleophryne, brachycephalids, Rhinoderma, and Allophryne) and Ranoidea, and all its descendants. Neobatrachian synapomorphies include the presence of a neopalatine bone (=palatine auctorum), fusion of the third distal carpal to other carpals (Ford, 1989b), the complete separation of the sartorius muscle from the semitendinosus, the presence of an accessory head of the adductor longus muscle, and the absence of the parahyoid bone (Cannatella, 1985; Duellman and Trueb, 1986; Dunlap, 1960).
Another likely synapomorphy is the presence of a 55-bp insertion in the 28S ribosomal RNA gene (Hillis and Davis, 1987). This is present in neobatrachians (including the limnodynastine Limnodynastes, the sooglossid Nesomantis, and the leptodactylid Telmatobius), but was not present in caecilians, salamanders, or other frogs, including pelobatoids.
Some recent classifications divided Neobatrachia into three superfamilies: Bufonoidea (=Hyloidea), Ranoidea, and Microhyloidea (Duellman, 1975; Laurent, 1986), but relationships among the clades within these groups are not clear.
There are no published synapomorphies of "Bufonoidea" (=Hyloidea, Dubois [1986]) as generally recognized (Duellman, 1975; Laurent, 1986); hyloids are simply neobatrachians that are not ranoids. In a phylogenetic analysis of osteological characters of Neobatrachia, Ford (1989b) found hyloids to be paraphyletic with respect to Ranoidea, including Microhylidae. However, Hay et al. (1995) presented a neighbor-joining tree of relationships among most of the families of frogs, in which the hyloids were monophyletic.
=======C====== Allophryne ruthveni
|
|============= Brachycephalidae
|
|============= Bufonidae
|
|======B====== Heleophryne
|
|||||||||||||| Leptodactylidae
|
|======D====== Limnodynastinae
|
| ====D== Myobatrachinae
|===G==|
| ======= Sooglossidae
===|
|======E====== Rhinoderma
|
| ======= Hylidae
| |
|===H==|====== Pseudidae
| |
| ======= Centrolenidae
|
| ======= Microhylidae
| |
| |===F== Hemisus
| |
| |====== Dendrobatidae
| |
====A==||||||| Arthroleptidae
|
||||||| Ranidae
|
|====== Hyperoliidae
|
======= Rhacophoridae
A hypothesis for the phylogenetic relationships of Neobatrachia.
Ranoidea Node A
Ranoids are a large and very diverse group. In a sense they are a radiation equivalent to the "hyloids" (=bufonoids). (Realize however that there is little evidence for hyloid monophyly, whereas there are data supporting ranoid monophyly, depending on the inclusion of Dendrobatidae.) Ranoids however are firmisternal, meaning that the left and right epicoracoid cartilages of the shoulder girdle are fused. In contrast, the hyloids are mostly arciferal, meaning that the cartilages overlap, and are not fused.
Ranoids can be viewed as the Old World counterpart of the hylids, bufonids, and leptodactylids. "Ranidae" are analogous to "Leptodactylidae" in being a diverse, paraphyletic group of non-treefrog forms, but with their center of diversity in Africa and Southeast Asia, with a minor radiation of the genus Rana in the New World. Rhacophoridae and Hyperoliidae are the Old World ranoid treefrog types, analogous to hylids in Central and South America. Microhylids are perhaps the firmisternal equivalent to Bufonidae in that they are a cosmopolitan group with three apparent radiations, in South America, Africa, and southeast Asia.
One major issue in the content of the Ranoidea is the inclusion (or not) of the Dendrobatidae (poison-dart frogs). Here we follow Ford (1989b) in treating dendrobatids as ranoids. Analysis of rRNA sequence data by Hay et al. (1995) does not support this, however. The four species of ranoids analyzed by Hay et al. (1995) formed a monophyletic group, but the dendrobatid was placed among the "hyloids."
The name Ranoidea was defined by Ford and Cannatella (1993) as the node-based name for the last common ancestor of hyperoliids, rhacophorids, ranids, dendrobatids, Hemisus, arthroleptids, and microhylids, and all of its descendants. Synapomorphies of Ranoidea include completely fused epicoracoid cartilages, and the medial end of the coracoid being wider than lateral end (Ford, 1989b). A probable synapomorphy, depending on the position of Dendrobatidae within Ranoidea, is the insertion of the semitendinosus tendon dorsal to the gracilis muscle (convergent with some myobatrachines).
Traditionally, Ranoidea was diagnosed by the condition of the pectoral girdle described as firmisterny (Duellman and Trueb, 1986). This feature has been controversial because of ambiguity in the definition of the term. One of the simpler and more widely accepted definitions of firmisterny is the complete fusion of the epicoracoid cartilages (Peters, 1964). The girdle has been treated as a single complex unit, and many individual aspects of it (i. e., clavicle, coracoid, epicoracoid and procoracoid cartilages) have been not used in systematic analyses (Ford, 1989a). The validity of the firmisternal girdle as a single character cannot be assessed until the individual elements are examined for character independence (Ford, 1988, 1989b).
Heleophryne Node B
The redundant family name applied to this taxon is Heleophrynidae.
Allophryne ruthveni Node C
The redundant family name applied to this taxon is Allophrynidae.
"Myobatrachidae" Node D
Under most arrangements, Limnodynastinae and Myobatrachinae are included in the family Myobatrachidae. The arrangement here follows Ford and Cannatella (1993).
Rhinoderma Node E
The redundant family name applied to this taxon is Rhinodermatidae.
Hemisus Node F
The redundant family name applied to this taxon is Hemisotidae.
Unnamed Node G
No name has been proposed for this node. Under Linnean nomenclature the name Myobatrachidae has priority over Sooglossidae.
Unnamed Node H
Lynch (1973) and Duellman and Trueb (1986:character J) used the presence of intercalary elements to unite Hylidae, Centrolenidae and Pseudidae into a clade. Intercalary elements are also known in Hyperoliidae and Rhacophoridae, as well as mantelline ranids and phrynomerine microhylids. The tree presented by Duellman and Trueb (1986:Fig. 17-3) has an equally parsimonious solution that would require convergent evolution of a firmisternal girdle (character C1) if lack of homoplasy in the intercalary element (character J1) is favored. In a subtree Duellman and Trueb (1986:Fig. 17-4) used the presence of intercalary elements to unite mantellines, hyperoliids, and rhacophorids, but not phrynomerines. This tree also has an equally parsimonious alternative that would unite phrynomerines with the aforementioned clade, but requires homoplasy in the Type 2 larva (character O2').
Ford's (1989b) study of dendrobatid relationships did not include centrolenids or pseudids. Ford and Cannatella continued the recognition of the clade consisting of Hylidae, Pseudidae, and Centrolenidae, which is diagnosed by the presence of intercalary elements, but did not name it. Hay et al. (1995) found that pseudids, hylids and centrolenids (each represented by one species) not to be a clade, although the latter two were sister-taxa, and all three were part of the Neobatrachia.
Recently, Ruvinsky and Maxson (1996) analyzed relationships among Neobatrachia based on mitochondrial 12S and 16S sequences. Their neighbor-joining tree is presented below. Several features of their tree are noteworthy.

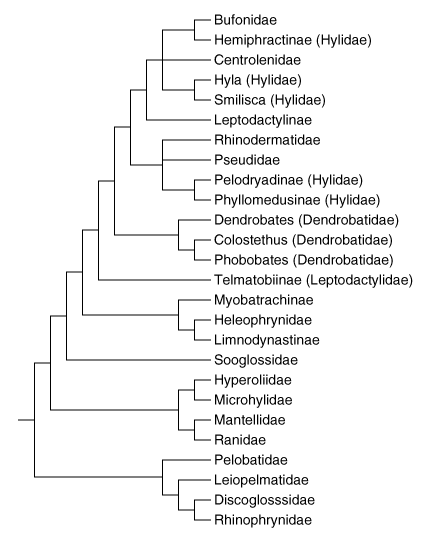
Phylogenetic relationships of Neobatrachia after Ruvinsky & Maxson 1996.
Bufonoids are a monophyletic group, and dendrobatids are nested within this clade. Ranoids are monophyletic also. Hylidae is polyphyletic, with some hylids being most closely related to Bufonidae, some to this clade plus Centrolenidae, and some to Pseudidae and Rhinodermatidae. Leptodactylidae as represented by Telmatobiinae and Leptodactylinae can be considered paraphyletic in one considers that Bufonidae, Hylidae, Rhinodermatidae, Pseudidae, and Dendrobatidae are all derived from Leptodactylidae, an arrangement that agrees with Lynch (1971).
References
Cannatella, D. C. 1985. A phylogeny of primitive frogs (archaeobatrachians). Ph.D. Dissertation, The University of Kansas, Lawrence.
Duellman, W. E. 1975. On the classification of frogs. Occ. Pap. Mus. Nat. Hist. Univ. Kansas (42):1-15.
Duellman, W. E., and L. Trueb. 1986. Biology of Amphibians. McGraw-Hill Book Co., New York.
Dunlap, D. G. 1960. The comparative myology of the pelvic appendage in the Salientia. J. Morph. 106:1-76.
Ford, L. S. 1989a. Phylogenetic assessment of the anuran pectoral girdle. Ann. Soc. Roy. Zool. Belg. 119 (Suppl. 1):101-102.
Ford, L. S. 1989b. The phylogenetic position of poison-dart frogs (Dendrobatidae): reassessment of the neobatrachian phylogeny with commentary on complex character systems. Ph.D. Dissertation, The University of Kansas, Lawrence, Kansas.
Ford, L. S., and D. C. Cannatella. 1993. The major clades of frogs. Herp. Monogr. 7:94-117.
Hay, J. M., I. Ruvinsky, S. B. Hedges, and L. R. Maxson. 1995. Phylogenetic relationships of amphibian families inferred from DNA sequences of mitochondrial 12S and 16S ribosomal RNA genes. Mol. Biol. Evol. 12(5):928-937.
Hillis, D. M., and S. K. Davis. 1987. Evolution of the 28S ribosomal RNA gene in anurans: regions of variability and their phylogenetic implications. Mol. Biol. Evol. 4:117-125.
Laurent, R. F. 1986. Sous classe des lissamphibiens. Systématique. Pp. 594-797 In P.-P. Grassé and M. Delsol (Eds.), Traité de Zoologie, Tome 14, 1B. Masson, Paris.
Lynch, J. D. 1971. Evolutionary relationships, osteology, and zoogeography of leptodactyloid frogs. Misc. Publ. Mus. Nat. Hist. Univ. Kansas (53):531-238.
Lynch, J. D. 1973. The transition from archaic to advanced frogs. Pp. In J. L. Vial (Ed.), Evolutionary Biology of the Anurans: Contemporary Research on Major Problems. University of Missouri Press, Columbia.
Peters, J. A. 1964. Dictionary of Herpetology. Hafner Publishing Co., New York.
Ruvinsky, I. and L. R. Maxson. 1996. Phylogenetic relationships among Bufonoid frogs (Anura: Neobatrachia) inferred from mitochondrial DNA sequences. Molecular Phylogenetics and Evolution 5:533-547.
Title Illustrations

| Scientific Name | Bufo marinus |
|---|---|
| Comments | cane toad |
| Specimen Condition | Live Specimen |
| Source | #1354003 |
| Source Collection | Bugwood Network/Forestry Images |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright | © Kevin Enge, Florida Fish and Wildlife Conservation Commission |
| Scientific Name | Hypsiboas punctatus |
|---|---|
| Location | lowland Amazon rain forest, Peru |
| Specimen Condition | Live Specimen |
| Copyright |
© Greg and Marybeth Dimijian

|
About This Page
David Cannatella

University of Texas, Austin, Texas, USA

Museum of Comparative Zoology, Harvard University, Cambridge, Massachusetts, USA

University of Texas, Austin, Texas, USA
Correspondence regarding this page should be directed to David Cannatella at
Page copyright © 1995 David Cannatella
 Page: Tree of Life
Neobatrachia. "Higher" Frogs.
Authored by
David Cannatella, Linda Ford, and Lori Bockstanz.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Neobatrachia. "Higher" Frogs.
Authored by
David Cannatella, Linda Ford, and Lori Bockstanz.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- Content changed 20 December 2008
Citing this page:
Cannatella, David, Linda Ford, and Lori Bockstanz. 2008. Neobatrachia. "Higher" Frogs. Version 20 December 2008 (under construction). http://tolweb.org/Neobatrachia/16987/2008.12.20 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site