Pterotrachea coronata
Roger R. SeapyIntroduction
Pterotrachea coronata attains the largest size (to 385 mm) among the Pterotracheidae and has the most streamlined body shape. The proboscis is long and pointed, terminating in a small buccal mass. The trunk is long and slender, with ventro-lateral folds of cutis anterior to the swimming fin that form a concave bib. The visceral mass is long and slender (the highest length to width ratio in the genus), and the basal part of its length is deeply imbedded in the posterior end of the trunk. The tail is large and laterally flattened, creating a broad surface area that aids in burst swimming, attributable to strong side-to-side flexion of the trunk and tail. During burst swimming, the proboscis is tucked into a ventral groove in the anterior one-half of the trunk created by the ventro-lateral folds of cutis. When viewed dorsally the eyes have a rectangular shape with a retinal base somewhat wider than the lens; shape similar to that in P. scutata. A small fin sucker is located on the mid-ventral surface of the swimming fin in males only. The geographical distribution is cosmopolitan in tropical to subtropical waters.
Brief Diagnosis:
A species in the genus Pterotrachea with the following characteristics:
- Body shape streamlined for burst swimming, produced by side-to-side flexion of the elongate trunk and the laterally flattened tail
- During burst swimming the slender, elongate proboscis is tucked into the ventral groove formed by the ventro-lateral folds of the cutis
- Eyes rectangular in dorsal view, with the retinal base somewhat wider than the lens
- Visceral mass slender and elongate
- Swimming fin sucker small and only in males
Characteristics
- Body morphology
- Body length greatest among the pterotracheids (to 385 mm)
- Streamlined body shape; trunk elongate and cylindrical, tail large and laterally flattened (see title illustration)
- Proboscis long, slender and cylindrical, tapering to a small buccal mass (see title illustration)
- Eye shape rectangular in dorsal view, with retinal base somewhat wider than the lens; very similar in appearance to that in P. scutata. In Hawaiian P. coronata the average eye length to width ratio = 2.2, decreasing from 2.5 in small to 1.9 in large individuals (Seapy, 1985)
- Visceral nucleus long and narrow (see photograph below and title illustration). The shape contrasts markedly with the shorter, tear-drop shape of the visceral nucleus in P. scutata and P. hippocampus (see the respective species pages). In Hawaiian P. coronata the ratio of length to width averages 5.6 and ranges from 4.1-7.2 (Seapy, 1985).
- Visceral nucleus imbedded in the cutis, with only the tapered, pointed end extending above the posterior end of the trunk
 Click on an image to view larger version & data in a new window
Click on an image to view larger version & data in a new window
Figure. Posterior portion of body in a female Pterotrachea coronata, viewed from the left side. Note the gills projecting dorsally and the long egg string that extends below the visceral nucleus toward the lower right. Note also the linear arrangement of the developing embryos in the transparent mucus sleeve (click on photograph for enlarged view). ©
- Swimming fin closer to the visceral nucleus than to the head, with the anterior end of fin just anterior to the midpoint of the trunk (see title illustration)
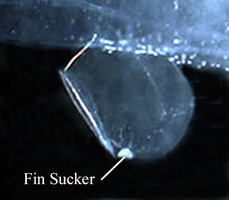
- Small sucker located medially on ventral margin of the swimming fin and present only in males
- Tail terminates in a pair of small, laterally-flattened lobes (see title illustration), between which emerges an elongate filament (see photograph below). In specimens collected by plankton nets and transferred to preservatives, the filament is usually missing; due either to damage during capture or subsequent detachment during preservation
- Shell present only in larvae and is lost following metamorphosis
- Larva unknown, but probably is represented by either larva 2 or larva 3 of Richter (1968); see the Pterotrachea page
- Radular morphology similar to that in the other species in the genus; the median cusp is conspicuously broader and longer than the lateral ones (see the Pterotrachea page)
Comments:
The largest individual recorded to date (365 mm) is attributable to a specimen collected at Shelly Beach, Bermaqui, New South Wales; as reported by Libby Hepburn in the Atlas of Life in the Coastal Wilderness (http://www.alcw.org.au/index.php/2012/12/biggest-carinaria-yet/). The estimated body length was determined from the meter tape in the photograph.

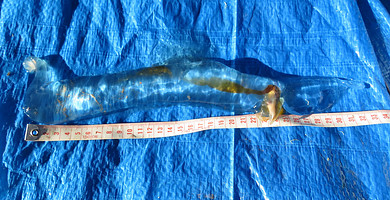
Figure. Largest recorded specimen of Pterotrachea coronata (OAL = 365 mm). The animal was washed ashore at Shelly Beach, Bermaqui, New South Wales, southeastern Australia. Additional photographs are available at http://www.alcw.org.au/index.php/2012/12/biggest-carinaria-yet/.
Swimming behavior has been observed in the field by R. Gilmer (cited in Lalli and Gilmer, 1989). Animals exhibited different behavior during daytime and nighttime hours. During the day animals swam with their ventral sides directed upward (see title illustration), which facilitates visual location of prey silhouetted against the lighted surface waters. Active swimming occurred when animals were observed pursuing prey or when they were disturbed by scuba divers (and, presumably, by "natural" predators). At other times they appeared to be neutrally buoyant, floating curled into loose balls. At night animals also appeared to be neutrally buoyant, but were oriented with their ventral sides directed downward with or without slow undulation of the swimming fin. In this position animals hypothetically could locate bioluminescent prey beneath them
The tail of Pterotrachea coronata is large and laterally flattened, creating a broad surface area that aids in burst swimming, which involves strong side-to-side flexion of the trunk and tail. During burst swimming, the proboscis is tucked into a ventral groove created by the ventro-lateral folds of the cutis in the anterior half of the trunk. An excellent photograph of an animal exhibiting burst swimming was obtained by Olivier le Corre while diving in Villefranche Bay in the Meditteranean Sea. Click on the photograph for an enlarged view, and note the right-side flexion of the body posterior to the swimming fin and the tail filament streaming posteriorly from between the tail lobes


Figure. In-situ photograph of Pterotrachea coronata swimming rapidly upward. © 1993 O. le Corre
Pterotrachea coronata was studied by Denton and Shaw (1961) to determine how these animals are able to achieve neutral buoyancy. The authors found that body fluids obtained by centrifugation of the gelatinous tissues had a lower density than sea water, attributable to the replacement of heavier sulfate by lighter chloride ions. The sulfate ion concentration in the body fluids was shown to be about three-fourths that in sea water.
Results from a study by Pafort-van Iersel (1983) of the diel vertical distributions of carinariids and pterotracheids along a north-south transect in the mid North Atlantic Ocean indicated that Pterotrachea coronata ranges through the epipelagic into the upper mesopelagic zone. The samples upon which the study was based were collected with paired 1-m2 and 8-m2 opening-closing, rectangular midwater trawls at depth intervals of 50-100 m, 100 m intervals between 100 and 500 m, and 500-1,000 m. Among the daytime tows a total of eight specimens were collected from 100-200 m, fourteen from 200-300 m, thirteen from 300-400 m, with none below 400 m. In the nighttime tows two individuals were recorded from 50-100 m, 52 from 100-200 m, twelve from 200-300 m, and none below 300 m. These results suggest a nocturnal vertical migration of about 100 m from a depth range of 200 to 400 m (the upper mesopelagic zone) during the day to 100-300 m at night. Because only two specimens were captured in the 50-100 m tows at night (with none collected during the day), the diel depth range of P. scutata appears to be mostly limited to depths below 100 m.
References
Denton, E. J. and T. I. P. Shaw. 1961. The buoyancy of gelatinous marine animals. Journal of Physiology, London 161: 14P-15P (Proceedings).
Lalli, C. M. and R. W. Gilmer. 1989. Pelagic snails. The biology of holoplanktonic gastropod mollusks. Stanford University Press, Stanford. 259 pp.
Pafort-van Iersel, T. 1983. Distribution and variation of Carinariidae and Pterotracheidae (Heteropoda, Gastropoda) of the Amsterdam Mid North Atlantic Plankton Expedition 1980. Beaufortia 33: 73-96.
Richter, G. 1968. Heteropoden und Heteropodenlarven im Oberflächenplankton des Golfs von Neapel. Pubblicazioni della Stazione Zoologica di Napoli 36: 346-400.
Richter, G. and R. R. Seapy. 1999. Heteropoda, pp. 621-647. In: D. Boltovskoy (ed.), South Atlantic Zooplankton. Backhuys Publishers, Leiden.
Seapy, R.R. 1985. The pelagic genus Pterotrachea (Gastropoda: Heteropoda) from Hawaiian waters: a taxonomic review. Malacologia 26(1-2): 125-135.
Title Illustrations

| Scientific Name | Pterotrachea coronata |
|---|---|
| Location | Hawaiian Islands |
| Specimen Condition | Live Specimen |
| Sex | Female |
| Life Cycle Stage | adult |
| View | left side |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
©

|
About This Page

California State University, Fullerton, California, USA
Correspondence regarding this page should be directed to Roger R. Seapy at
Page copyright © 2010
 Page: Tree of Life
Pterotrachea coronata .
Authored by
Roger R. Seapy.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Pterotrachea coronata .
Authored by
Roger R. Seapy.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 18 August 2008
- Content changed 17 July 2010
Citing this page:
Seapy, Roger R. 2010. Pterotrachea coronata . Version 17 July 2010 (under construction). http://tolweb.org/Pterotrachea_coronata/28738/2010.07.17 in The Tree of Life Web Project, http://tolweb.org/










 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site