Table of Contents
- Viruses - Where Do They Belong?
- The First Split
- DNA - RNA Reverse Transcribing Viruses
- Retroviridae
- Being Unique - the Mechanism That Sets Them Apart
- Lentivirus and the Subgenus Primate Serogroup
- Human Immunodeficiency Virus
- Evolution and Survival of Retroviruses - Implications to Humans and Healthcare
- Conclusion
Viruses – Where Do They Belong?
At the base of the tree of life, the roots, there are three proposed major lineages: eubacteria (the “true bacteria”), eukaryotes (which include protists, plants, fungi, animals, etc.) and archaea (the extremophiles such as methanogens, halophiles, sulfolobus and relatives).
With the discovery of viruses and the significant role they play in evolution, came one of the most controversial questions when studying the evolution of life: where do you place viruses? Unable to detect a fossil virus as a particle due to the size and fragility, studying viruses has been limited to the ones isolated in the present or at most a few decades old. As a result, studying the origins of viruses has proved to be difficult.
Placement in one of the three major branches of living organisms has been difficult because of questions of whether or not viruses are living organisms. Being parasitic, there exist viruses of all major classes of organisms – animals, plants, fungi, bacteria and archaea. It has been postulated that viruses evolved with their hosts in the seas and during the emergence of life from the seas to land, viruses also probably emerged from the water with their hosts. As a result, the three major branches do not share virus types between them, as divergence occurred so long ago.
The First Split
Viruses can be further divided into the following groups based on the mechanism of replication, the latter of which is the focus of this investigation.
- double-stranded RNA viruses
- single-stranded negative sense RNA viruses
- single-stranded positive sense RNA viruses
- single-stranded DNA viruses
- double-stranded DNA viruses
- DNA-RNA reverse transcribing viruses
DNA-RNA Reverse Transcribing Viruses
Viruses belonging to this group are differentiated by the mechanism stated in their name. Reverse transcribing viruses are viruses that transcribe their genetic information from RNA to DNA. Reverse transcribing viruses are further divided into 5 families. Hepadnaviridae and Caulimoviridae are families that are double-stranded DNA viruses which use an RNA intermediate during replication. Family Metaviridae are a family of viruses that exist as retrotransposons in a eukaryotic host’s genome. Family Pseudoviridae infect fungal or invertebrate. The latter two families and Retroviridae are single-stranded RNA viruses, which use a DNA intermediate to replicate.
Retroviridae
This family of viruses is a large and diverse family that is defined by common structure, composition, and replicative properties. First discovered in 1908 by Vilhelm Ellermann and Oluf Bang, members of this family replicate via a DNA intermediate using a single stranded RNA genome. They can be broadly divided into two categories – simple and complex – based on the organization of their genomes. Being the most abundant group of subcellular parasites, they infect animals, plants and bacteria – and are behind severe diseases such as hepatitis, yellow fever and the infamous human immunodeficiency virus (HIV).
In general, virions are 80-100 nm in diameter, having an outer lipid envelope that incorporates and displays the viral glycoproteins. The single-stranded, linear RNA is found to be 7-12 kb in size. All retroviruses contain three important coding domains:
- gag - contains information to direct the synthesis of internal virion proteins that form the matrix, the capsid and the nucleoprotein structures
- pol - contains information for the reverse transcriptase and integrase enzymes
- env - contains the information for building the surface and transmembrane components of the viral envelope protein
Another significant coding domain present in this family is pro – a domain that encodes the virion protease. The differentiation between simple and complex retrovirus comes from the information that is carried in the genome. Simple retroviruses carry the above mentioned regions in their genome, whereas, complex retroviruses code for additional regulatory non-virion proteins derived from multiply spliced messages. Except for deltaretrovirus, lentivirus, and spumavirus, which are considered to be complex, the other four are simple retroviruses.


A) A simple retroviral genome containing the four major coding regions, gag, pro, pol, and env. B) A complex retroviral genome that contains, besides the major coding domains, information for two regulatory proteins, Tax and Rex © 1997 by Cold Spring Harbor Laboratory Press
Being Unique - the Mechanism That Sets Them Apart
Retroviruses have a unique mechanism, which not only accounts for the diversity of the viruses belonging to this group, but also for the success of group.
Upon entering the cell, the single-stranded RNA and reverse transcriptase are released from the virion envelope. Mediated by the host-cell RNA polymerase II, upon the action of reverse transcriptase, the ssRNA is converted to double-stranded DNA and incorporated into the host’s genome as a provirus. The virus now relies almost entirely on the host-cell machinery for the expression of the viral DNA. This viral DNA is then transcribed to produce a messenger RNA (mRNA). This mRNA is processed and then translated into a long polyprotein, which is cleaved by the protease to yield the individual proteins that make up the virion. The full-length mRNA have two functions – they encode for the gag and pol gene products and they are packaged into the virion particles as the progeny genomic RNA.
Lentivirus and Subgenus Primate Serogroup
Lentivirus (lenti– Latin for “slow”) is the genus of which human immunodeficiency virus belongs to. A complex genus from the Retroviridae family, Lentivirus is classified by the long incubation period of the virus. Thus far, five subgenus species have been classified. The subgenus of focus for this investigation is primate serogroup – to which besides HIV, simian immunodeficiency virus belongs to. This subgenus has been classified by the use of CD4 protein as a receptor and for the absence of dUTPase.
Human Immunodeficiency Virus
Transmission & Prevention
The source of infection is from bodily fluids of the carrier such as semen or blood. HIV can be transmitted through sexual activity, sharing used needles or syringes, mother-to-child transmission through childbirth or breast feeding or receiving transfusions of infected blood, transplanted organs or donated sperm. Entry into the target cells of the body occurs through a break in the skin, absorption through mucosal membranes (such as the mouth or eyes) or disruption through the mucosa. In order to prevent the transmission of this virus, one should take care to avoid the situations listed above.
Symptoms
HIV enters the body and infects cells in the immune system and central nervous system. T helper lymphocyte cells are immune system cells that coordinate the actions of other immune system cells. HIV infects this type of cell.
HIV attaches on to a protein found on the surface of the T helper cell – CD4. As mentioned before, primate serogroup – the subgenus to which HIV and SIV belong to, is classified by the use of CD4 protein as a receptor. Using these cells, it makes copies of itself and kills the CD4+ lymphocyte cells it infects. These cells are made by the body to help you fight infections – thus by reducing the number of CD4 cells in your body, you are more prone to infections.
Stages of HIV infection has been broken down into four steps: primary infection, clinically asymptomatic stage, symptomatic HIV infection, and progression from HIV to AIDS.
During stage 1, there is a short flu-like illness that is often mistaken for being nothing serious, thus diagnosis is frequently missed. This stage generally lasts for a few weeks, during which the immune system attempts to fight back by producing HIV antibodies and cytotoxic lymphocytes – known as seroconversion.
Stage two can last for an average of ten years and is free of major symptoms. Remember, HIV belongs to the Lentivirus family – distinguished by the long incubation period. HIV is not dormant however; it is active in the lymph nodes and this is displayed by swollen glands.
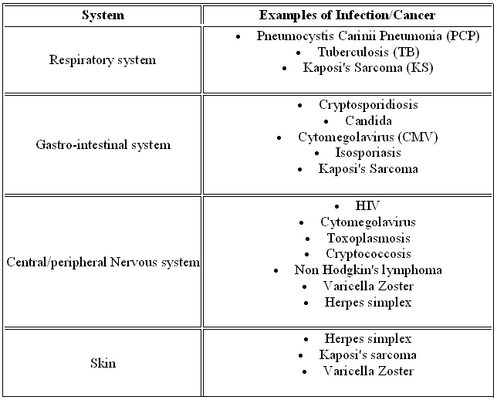
The symptomatic HIV infection stage occurs when the immune becomes severely damaged by HIV. The body has been fighting back for years, the virus could mutate and become stronger and the T helper cells are dying off, with turnover rates failing to keep up. Because of the damaged immune system, opportunistic infections and cancers occur. The table on the left has a list of common examples. The AIDS diagnosis occurs when the immune system becomes more and more damaged and results in severe illnesses. There are many clinical symptoms that are used to diagnosis which stage the patient is in. The table on the right lists some of these symptoms.
Treatment Options
Treating an opportunistic infection in an HIV patient is similar to treating HIV negative patients. However, the goal of many researchers today is to come up with an HIV antiretroviral treatment. These drugs attack the virus in four different ways by inhibiting steps in the mechanism of HIV.
The four different ways are nucleoside reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (featured on the right) and fusion or entry inhibitors. NRTIs and NNRTIs work by interfering with the action of reverse transcriptase. Protease inhibitors inhibit protease – which cleaves the polyprotein to yield active virions. Fusion or entry inhibitors prevent HIV from entering human cells. However, not only one drug is used to treat HIV patients; instead a combination of three or more drugs is used.
Evolution and Survival of Retroviruses – Implications to Humans and Healthcare
RNA viruses possess great genetic variability, which results in a rapid evolution. The RNA virus variations is a result of molecular mechanisms such as mutations, homologous and non-homologous recombination, recombination and genome re-assortment. Mutation rates during RNA genome replication and retro-transcription are much higher than the error rates found during the replication of DNA in eukaryotic and prokaryotic cells. Double-stranded DNA comes with a mismatch repair system that RNA viruses do not have. Whereas, most mutations can be caught by this repair system in DNA, RNA just carries forth the mutation. However, it has been postulated lack of this repair system has allowed RNA viruses to be more fit, allowing them to have mutations and adapt. Therefore, RNA viruses would probably still aim for high mutation rates.
How is this relevant to HIV and healthcare for humans? Due to the high mutation rates, drugs that are developed to fight against this virus become useless as time progresses. The drug will appear to work for some time, while virions in the host are mutating and building resistance against the drug. Eventually, the virions that the drug works successfully against will have died out, leaving only the virions that are resistance to once again, replicate and spread. As a result, this high mutation rate is kept in mind, as drugs are now being designed that are mutation friendly. However, this is proving to be difficult as accounting for mutations reduces the favourability of the drug to specific target.
Conclusion
Understanding retroviruses and their mechanisms is crucial to the humans, as many people around us are affected by these viruses. Human immunodeficiency virus is causing an epidemic in the present and studying the evolutionary history of these viruses can allows us to develop a defensive strategy for those suffering from AIDS and other diseases caused by viruses.
Studying Retroviruses opens up new areas of interest it not only biology, medicine, and healthcare, but also in biotechnology. Studying the mechanism of replication by these retroviruses, have allowed scientists studying the origins of life on earth to postulate that perhaps it was by this same mechanism that life originated: the RNA world theory. As this theory states, rather than RNA molecules being formed from DNA, it has been posited that DNA was transcribed from RNA, similar to the mechanism that these viruses carry out. These viruses have been useful for studying genetic information and cellular growth control. In terms of biotechnology, these viruses have provided essential tools – allowing for the generation of cDNA copies of RNA that are manipulated for cloning and sequencing. There are future hopes in using these viruses as delivery vehicles for human gene therapy. There is a promising future in the retroviruses.








 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site